- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

Before you read on, you might want to download this entire revision notes in PDF format to print it out for your child, or yourself to read it later.
This will be delivered to your email inbox.
What does it mean to be living? Officially, living organisms are biological entities that exhibit life processes such as growth, reproduction, response to stimuli and metabolism. However, where do we draw the line for this definition? As we know, viruses hijack their host cells and use their machinery for their own replication. Could this still be considered replication, and can we still consider viruses as living? As we have seen in the latest pandemic, viruses are also more than capable of rapid mutation and evolving adaptations such as better infectivity or severity.
In this article, we will cover the general structure of viruses, and some examples of bacterial and animal cell-infecting viruses.
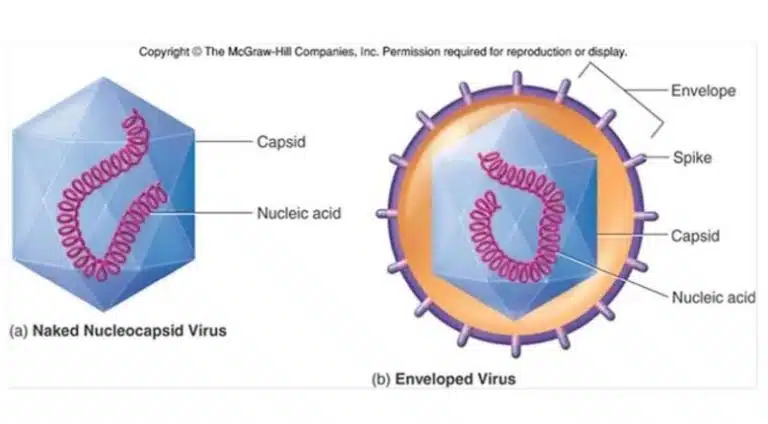
By definition, viruses are small obligate intracellular parasites, which contain either a RNA or DNA genome surrounded by a protective, virus-coded protein coat. The protein coat is known as a capsid, and encloses the viral genome along with several essential proteins. Viruses that infect animal cells tend to have an additional envelope, consisting of a lipid membrane derived from the host cell from which it buds off from. The envelope is also embedded with viral proteins which help with host cell recognition.
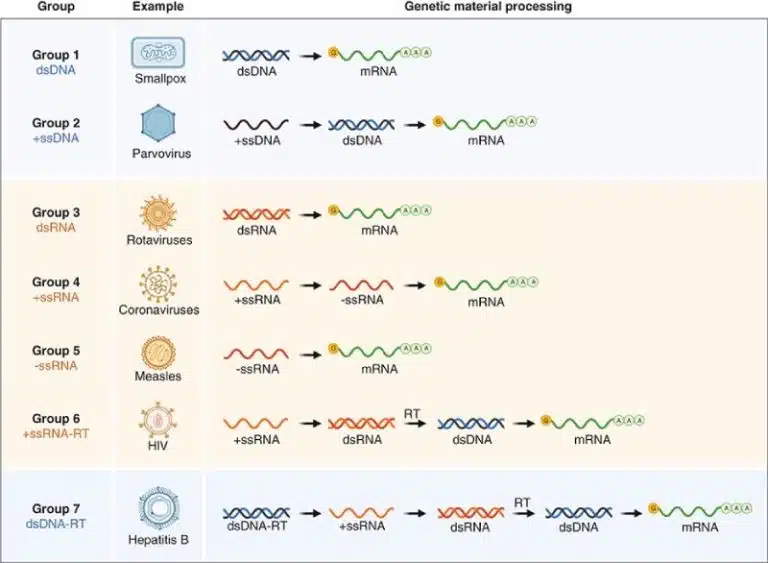
Viruses come in many shapes and forms, so how can we classify them? Recall the central dogma of biology, where the genetic sequence to be translated into protein is encoded within mRNA. Hence, viruses have to convert their genetic material into mRNA one way or another, so that it can be translated into functional protein by the host cell’s machinery.
The Baltimore classification system, developed by David Baltimore, categorized viruses based on their genome type and how they produce mRNA. On top of being a neat and nifty criteria for classifying such a diverse group of organisms, the Baltimore classification system is also crucial for understanding viral replication strategies.
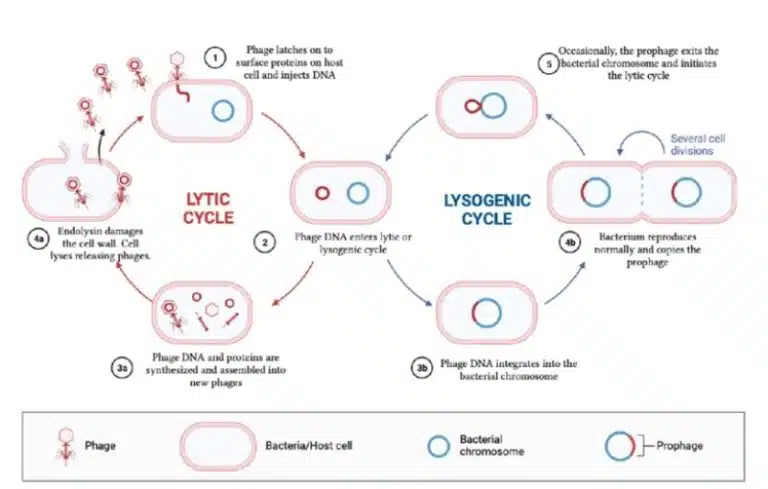
Let us look at two examples of viruses that infect bacteria, known as bacteriophages. After infecting its bacterial host, bacteriophages enter one of the two main pathways of viral replication, namely the lytic cycle and lysogenic cycle.
In the lytic cycle, the phage infects a host bacterium and immediately uses the host’s machinery to make copies of its genome and synthesize its proteins. This process results in rapid production of new viruses, which eventually results in lysis of the host cell.
In the lysogenic cycle, the viral genome is integrated into the host bacterium’s chromosome. As the bacterium undergoes DNA replication and cell division, the viral genome is replicated and passed down to daughter cells as well. The phage hence remains dormant and can be triggered to switch back to the lytic cycle under certain conditions such as stress or environmental signals. Staying in the lysogenic cycle allows the phage to survive and spread amongst the host’s daughter cells, without immediately killing the host.
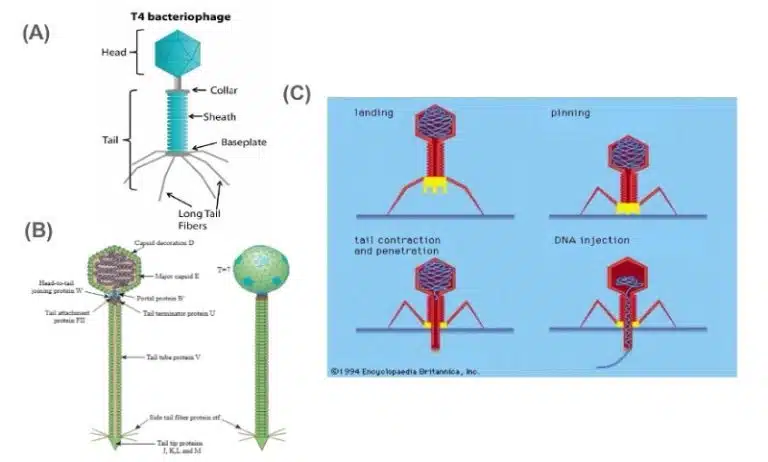
An example of a phage that replicates through the lytic cycle is the T4 phage. The T4 phage (Escherichia virus T4) is a species of bacteriophages that infect Escherichia coli bacteria. Its genome consists of dsDNA packed into an icosahedral head. The capsid consists of a head and tail portion, along with long spider leg-like tail fibers at the base of the phage (Fig 4A). The T4 phage only undergoes the lytic cycle during its replication.
Attachment: When it encounters its host cell, tail fibers bind to complementary receptors on the host cell surface.
Entry: The phage secretes lysozyme, increasing the permeability of the host cell wall. The tail portion of the phage’s capsid contracts, and phage DNA is injected into the host cell.
Replication: Once within the host cell, host cell machinery such as helicases, DNA polymerases and RNA polymerases are used to generate more copies of the viral dsDNA genome, along with transcription of the viral genome into mRNA. The viral mRNA encodes all the proteins that are required to synthesize new phages, and host cell ribosomes are used to translate viral mRNAs into protein. New phages are synthesized by packing newly synthesized viral genomes within capsids composed of newly synthesised viral proteins.
Exit: The rapid production of phages eventually results in the lysis of the host cell, releasing new T4 phages into the surroundings. These phages then continue to find their new hosts, starting the cycle of viral invasion and replication all over again.
The lambda phage also infects Escherichia coli, but is able to enter the lysogenic cycle unlike the T4 phage. Structurally, the lambda phage also shares some similarities with the T4 phage, like having a dsDNA genome enclosed within the head of the capsid, having a tail sheath and even some tail fibers.
The process of attachment and entry for the lambda phage are the same as that of the T4 phage. However, instead of immediately using the host cell’s machinery to replicate itself, the lambda phage enters the lysogenic cycle, where its genome is integrated into the host cell’s genome using the viral enzyme integrase and replicated along with the host cell. The integrated phage DNA is known as a prophage. While existing as a prophage in the lysogenic cycle, the expression of viral proteins is repressed, hence new phages are not being synthesized.
Under certain conditions, usually involving host cell stress, host cell proteases are activated and degrade the proteins repressing expression of viral genes. The prophage is excised from the bacterial genome and expressed. At this point, the lambda phage switches over to the lytic cycle, and new lambda phage particles are synthesised similar to that of the T4 phage (see previous section on T4 replication and exit).
Animal-infecting viruses tend to come with an additional envelope enclosing the nucleocapsid. These envelopes are studded with viral glycoproteins that recognise their host cells and initiate invasion.
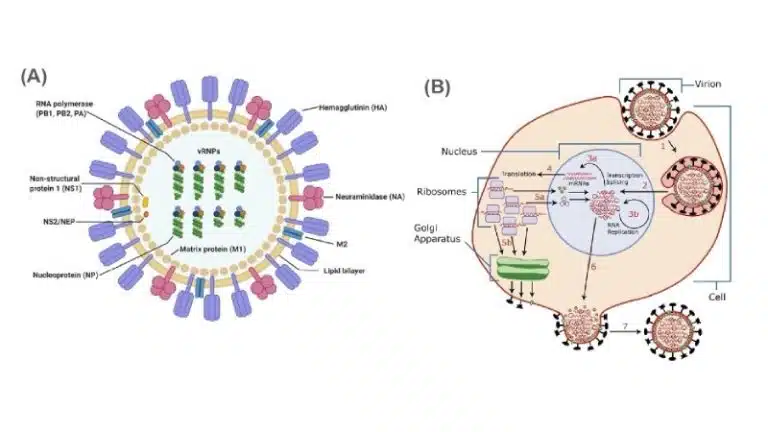
Influenza, commonly known as the flu, is a contagious respiratory illness caused by the influenza virus. There are several types, namely Influenza A, B, C and D. Influenza A is the most common and can be further classified into subtypes based on their surface proteins H (hemagglutinin) and N (neuraminidase) (eg. H1N1 and H5N1).
The influenza virus primarily infects epithelial cells in the respiratory tract. On its envelope, it carries the glycoproteins hemagglutinin and neuraminidase which are important for viral attachment and exit respectively. Within its capsid, the influenza virus carries 8 negative sense single stranded RNA (-ssRNA) and RNA-dependent RNA polymerase.
Attachment: Hemagglutinin on the viral envelope binds to sialic acid receptors on the host cell.
Entry: The virus enters the host cell through endocytosis. Lysosomes fuse with the endocytic vesicle holding the virus, and the subsequent decrease in pH results in the release of viral capsid into the cellular cytosol. Once in the cytosol, capsid proteins are degraded to release the 8 -ssRNA strands and RNA-dependent RNA polymerase, allowing them to reach the nucleus of the host cell.
Replication: In the nucleus, RNA-dependent RNA polymerase transcribes -ssRNA into +ssRNA. The sequence of -ssRNA is complementary to that of the coding sequence (non-coding). On the other hand, +ssRNA can be directly translated as mRNA to produce viral proteins. +ssRNA also serves as the template for RNA-dependent RNA polymerase to generate more copies of the viral genome, by generating more -ssRNA.
+ssRNA is translated outside the nucleus to synthesize viral proteins using host ribosomes. Capsid proteins are synthesized in the cytosol, while envelope proteins (hemagglutinin and neuraminidase) are synthesized at the rough endoplasmic reticulum. The envelope proteins undergo the secretory pathway similar to other membrane proteins, being trafficked to the golgi and finally being embedded into the host cell membrane.
Within the cytosol, the newly synthesized viral genomes and proteins (RNA-dependent RNA polymerase) are packed into the capsid.
Exit: Newly formed capsids exit the host cell through budding. During the process, they gain their envelope derived from host cell phospholipids, which are studded with hemagglutinin and neuraminidase. As the host cell membrane contains sialic acid receptors which are what binds to hemagglutinin in the first place, neuraminidase facilitates the release of these new viral particles by cleaving sialic acid from the surface of the host cell.
It is recommended for one to take their flu jab annually. As we have seen from influenza pandemics like the Spanish flu, Bird flu and more recently Swine flu, the influenza virus is extremely capable of rapid mutation and constant evolution. Hence, it is important for scientists to constantly be on their toes and monitor the global influenza situation, so as to keep ourselves ready and protected against the most commonly circulating strains.
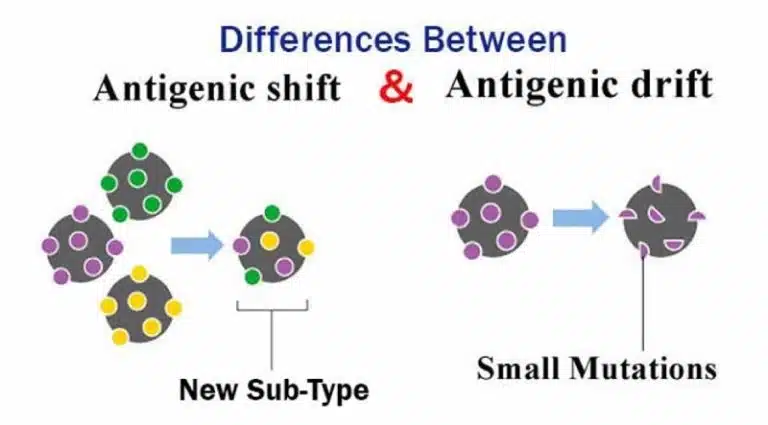
Antigenic drift and antigenic shift are two ways influenza viruses can change their envelope proteins, allowing them to evade the host’s immune system. These changes are caused by mutations, which result in changes in protein structure and binding of these envelope proteins to their host cell targets.
Antigenic drift involves small and gradual changes (due to mutations), leading to minor changes in the structures of hemagglutinin and neuraminidase. On the other hand, antigenic shift occurs when there is a sudden and major change in envelope proteins. This could happen due to a reassortment and shuffling of viral RNA segments. For example, if two or more viruses infect the same cell, their RNA segments can be shuffled, leading to new viruses with a unique combination of surface antigens. Such a new combination can be deadly and since most people would not have pre-existing immunity against this new strain, it could potentially lead to a pandemic.
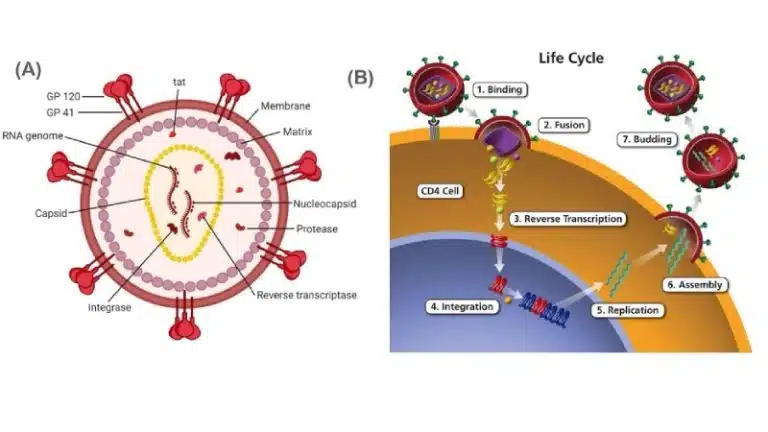
Human immunodeficiency virus (HIV) is a retrovirus that infects human immune cells (particularly CD4+ T cells, dendritic cells and macrophages). HIV infections are characterised by low CD4+ T cell counts. Especially without proper antiretroviral medications, the immune system gets increasingly compromised, eventually leading to acquired immunodeficiency syndrome (AIDS), the progressive failure of the immune system that allows life-threatening opportunistic infections and cancers to thrive.
The HIV virus, like the influenza virus, also consists of a nucleocapsid enclosed within a host cell membrane-derived envelope. The nucleocapsid contains 2 identical copies of the +ssRNA viral genome, along with essential viral proteins such as reverse transcriptase, integrase and protease.
Attachment:
gp120, which is attached to the viral envelope through gp41, binds to CD4 receptors on the surface of immune cells.
Entry: The viral envelope fuses with the host cell’s membrane, releasing the nucleocapsid into the cytosol of the host cell. The capsid is degraded and the viral genome and proteins are released into the cytosol.
Replication: The +ssRNA genome is converted to dsDNA using reverse transcriptase. This newly synthesized DNA enters the nucleus, where it is incorporated into the host cell genome using integrase. The integrated viral DNA is known as a provirus, which will remain latent for an extended period of time.
When activated, the provirus DNA is transcribed into mRNA which enters the cytosol for translation. The viral mRNA is translated as a polyprotein (long single chain of polypeptide containing multiple proteins) and has to be cleaved by protease into individual functional proteins. The viral +ssRNA genome is also transcribed from the provirus DNA sequence.
Exit: The viral genome and proteins are packaged into nucleocapsids, and the nucleocapsids exit the host cell through budding, gaining their envelopes. Similar to the influenza virus, viral surface proteins have undergone the secretory pathway and are embedded into the host cell surface, and are hence incorporated into the viral envelope as the virus leaves the host cell.
Even though viruses have caused living beings much grief and disease, scientists have found ways to put their abilities to good use. Here are some interesting applications of the viruses we’ve covered in this article!
The rampant use of antibiotics have resulted in an age of antibiotic resistance. One alternative to indiscriminate antibiotic usage is phage therapy, where phages are used to treat pathogenic bacterial infections. This therapeutic approach has been in the making since the early 20th century, but was progressively replaced by antibiotic usage following WW2.
In light of rising antibiotic resistance, interest in phage therapy has been making a comeback in recent decades. Due to its specificity, phages only target specific bacterial strains, minimizing harm to beneficial bacteria in one’s microbiome. Additionally, phage therapy can be customized to address individual bacterial infections, and can even be combined with antibiotics to target antibiotic-resistant bacteria.
However, phage therapy is largely still in development and not widely approved as a standard treatment in most countries. There is still much research and development to be done before phage therapy may become as commonplace as standard antibiotic treatment is today.
HIV is a species of lentivirus, a class of virus that integrates their genome into their host cell as proviruses, resulting in chronic and deadly diseases in humans and other mammalian species. However, it is exactly the lentivirus’ ability to integrate a large fragment of DNA into its host cell so stably over a lifetime, that scientists wish to harvest and use it for good.
Lentiviruses are used as vectors for gene therapy to introduce genetic material into cells, particularly to treat genetic diseases and cancers. Lentiviruses can integrate their genetic material very stably into their host cell’s genome, allowing for long-term expression of the introduced gene. Lentiviruses can also deliver large DNA sequences to both dividing and non-dividing cells, which is crucial for treating a wide range of conditions. As scientists continue to work on and improve the lentiviral vector, modern lentiviral vectors are designed to be safe, greatly reducing risks of any adverse side effects. So far, lentiviral vectors have entered various clinical trials against diseases like severed combined immunodeficiency (SCID), hemoglobinopathies and cancers.
We’ve learned about how HIV uses reverse transcriptase to convert +ssRNA into DNA. Scientists have figured out how to purify reverse transcriptase for use in research, and now we are able to convert mRNAs into complementary DNAs (cDNA) in-vitro! The reverse transcriptase enzyme is crucial for many research techniques (such as RT-PCR, which was used in COVID19 detection) and allows scientists to study transient mRNAs by converting them to the more stable cDNA.
Despite the devastating effects of viruses, we must remember that viruses have been around and evolved alongside us, for just as long as living beings have been around. They may be simple creatures that require a host to replicate, but their rate of mutation, evolution and adaptation has always been remarkably efficient, allowing them to constantly be on top of the living beings they need. At this point, to what extent can we agree that viruses are non-living beings?
Find out more by joining us at Science of Studying!
Prepared by: Michelle
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us