- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

This article is written based on the 9729 Chemistry GCE Advanced Level H2 Syllabus.
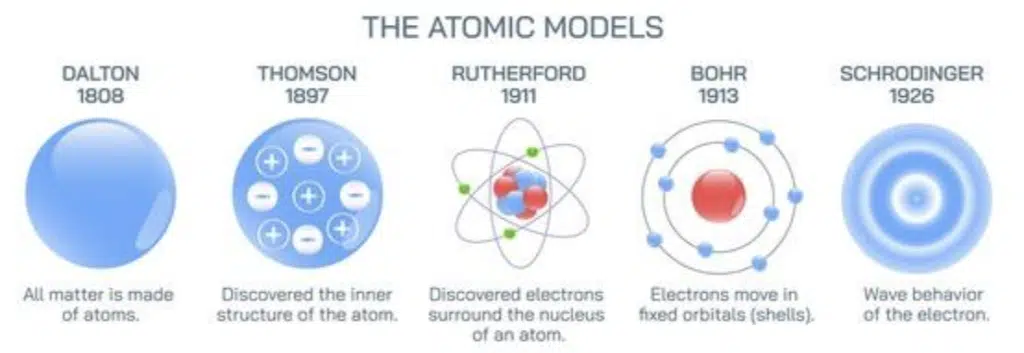
In secondary school, we have learnt atoms are the smallest unit of matter and are the building blocks of everything around us. We have also learnt that atoms are made up of even smaller components: sub-atomic particles which include protons, neutrons and electrons. Although it now seems trivial and obvious to us, our modern understanding of atoms is in fact over two millennia in the making, and is still being refined to this day.
The idea of an indivisible unit of matter dates back to the Ancient Greeks. The philosopher Democritus and his mentor Leucippus are often credited with proposing the earliest theory of an atom around 430 BC, and for naming them atomos, meaning ‘indivisible’. They imagined atoms as indivisible spheres of different sizes, with different properties corresponding to the type of matter they made up – for example, water atoms were proposed to be smooth and slippery.
This early philosophy of atoms went largely ignored by scientists until 1803, when English chemist John Dalton built upon it to develop a more scientific model of atoms. Dalton retained the ideas that atoms were small, indivisible spheres of matter, and atoms of a given element were identical to each other. He also added on that during chemical reactions, different atoms combine in fixed whole number ratios to form what we now call molecules.
But before you go on reading… You might want to download a pdf copy of this article as it is quite long!
Click the ‘Download’ button, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)


Outside of chemistry, Dalton was one of the first scientists to study colour-blindness, observing that it must be genetic as both him and his brother were red-green colourblind.
In 1897, J. J. Thomson discovered the electron, and through his cathode-ray experiments found that the negatively-charged particle was in fact present within atoms as well. He challenged the idea of an atom as an indivisible sphere, and in 1904 proposed what would later be commonly known as the ‘plum pudding’ atomic model, with negative electrons distributed within a sphere of positive charge.

Joseph John “J.J.” Thomson was also a teacher, and many of his students, as well as his own son George Paget Thomson, would go on to win Nobel Prizes of their own.
A few years later, his own student Ernest Rutheford ended up disproving this theory, when his team found that when positively-charged alpha particles were fired at a sheet of gold foil, although the majority of them passed through with little to no deflection, a very small number of alpha particles were deflected at huge angles. This implied that the majority of the atom was in fact empty space, with a very dense core of positive charge (later known as the nucleus) that repelled the alpha particles. Rutherford proposed that the electrons in Thomson’s model actually orbited the nucleus, but was unable to explain why they remained in orbit instead of losing energy and spiralling into the nucleus.
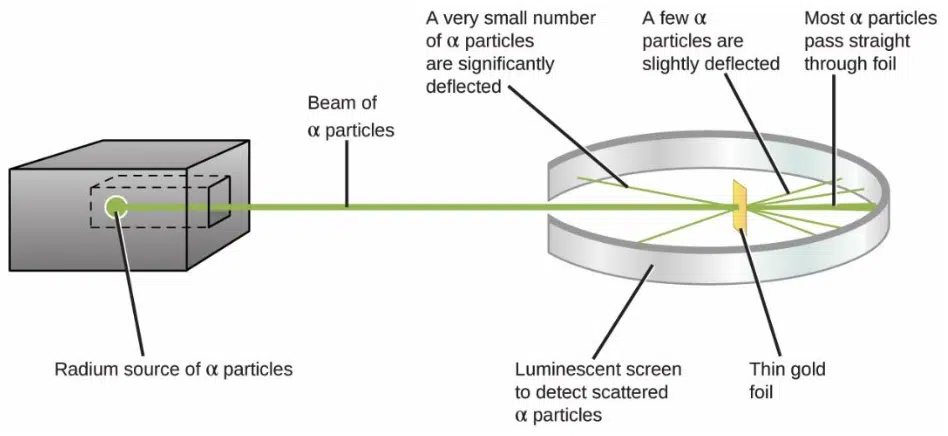
The results of Rutheford’s experiments so defied his expectations that he famously commented that it was “as if you fired a 15-inch [artillery] shell at a piece of tissue paper and it came back and hit you.”
In 1913, Danish physicist Neils Bohr proposed a solution to this problem by theorizing that electrons in an atom could only occupy orbits of fixed sizes, as only certain discrete energy levels were allowed in the atom. Electrons could move between these discrete energy states by absorbing or emitting light of specific wavelengths. Bohr’s model was the first to incorporate quantum physics, then a very new and emerging field of study, into atomic theory, and could predict the energy and emission spectra of single electron systems with high accuracy.
Although it cannot explain the emission behaviour of larger atoms, and eventually needed further refinement due to later breakthroughs in quantum physics, Bohr’s model plays an important part in chemistry education to this day, as it can be used to explain valency, chemical bonding and reactivity of atoms at a simple level.
Development of Schrodinger’s quantum model
In 1924, Louis de Broglie postulated that all particles, including atoms and electrons, could in fact be treated as matter waves with a specific wavelength, and attempted to explain the fixed energy levels in Bohr’s model by suggesting that the orbits were in fact standing waves, and hence only orbits with circumferences that are integer multiples of the wavelength of an electron are possible.
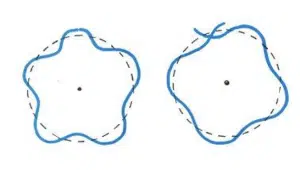
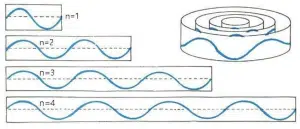
de Broglie’s hypothesis of electron orbits as “standing matter waves” – only orbits which exactly fit an integer number of wavelengths can be allowed to exist without collapsing due to destructive interference.
However, the concept of wave-particle duality also implies an inherent limitation to measuring certain properties of an electron at the same time. This will later be known as Heisenberg’s Uncertainty Principle, and it states that it is impossible to know the exact position of an electron and its exact momentum at the same time – a limitation which Bohr’s model violates.
In 1926, Erwin Schrodinger refined de Broglie’s hypothesis by applying the idea of electrons as standing waves to a three-dimensional space rather than a circular orbit. He solved a series of mathematical equations (known as Schrodinger’s equations), the solutions to which gave functions that described areas of space around the nucleus where electrons were probabilistically likely to be found, called atomic orbitals. Using this model, atoms could be described as nuclei surrounded by electron clouds with negative charge, and areas where electrons are more likely to be found can be said to have high electron density.
The quantum model is consistent with Heisenberg’s Uncertainty Principle, as orbitals only describe areas with 90% probability of finding an electron, without suggesting the exact position of the electron at any specific time.
The quantum model of atoms builds upon Bohr’s model in stating that electrons in an atom can only occupy discrete energy levels, which is labelled by a principal quantum number n (corresponding to electron shell number n in Bohr’s model). However, instead of orbiting the nucleus, the position of electrons is described probabilistically, with a 90% chance of being found within a 3D region in space around the nucleus called an orbital. Each orbital is described by a set of three quantum numbers:
In addition, each electron in the atomic system has a fourth quantum number, known as spin number (ms) which can take on the value of either +½ or −½.
Quantum Number | Symbol | Possible Values | Physical Interpretation |
principal | n | 1, 2, 3, 4, … | quantum shell, size of orbital |
angular momentum | ℓ | 0 ℓ n−1 | subshell, shape/type of orbital |
magnetic | mℓ | −ℓ mℓ ℓ | orientation of orbital |
spin | ms | +½ or −½ | upward or downward ‘spin’ of electron |
Summary of quantum numbers and their meanings
No two electrons in an atom may have the exact same set of quantum numbers ─ this is known as Pauli’s Exclusion Principle. This limits the number of electrons that can occupy the same orbital to two – one with an upward spin, one with a downward spin.
Principal quantum shell | Subshells | Orbitals | Number of electrons in subshell | Maximum number of electrons |
n = 1 | s (ℓ = 0) | 1s (mℓ = 0) | 2 | 2 |
n = 2 | s (ℓ = 0) | 2s (mℓ = 0) | 2 | 8 |
p (ℓ = 1) | 2px, 2py, 2pz (mℓ = −1, 0, 1) | 6 | ||
n = 3 | s (ℓ = 0) | 3s (mℓ = 0) | 2 | 18 |
p (ℓ = 1) | 3px, 3py, 3pz (mℓ = −1, 0, 1) | 6 | ||
d (ℓ = 2) | 3dxy, 3dyz, 3dxz, 3dz2, 3dx2–y2 (mℓ = −2, −1, 0, 1, 2) | 10 | ||
n = 4 | s (ℓ = 0) | 4s (mℓ = 0) | 2 | 32 |
p (ℓ = 1) | 4px, 4py, 4pz (mℓ = −1, 0, 1) | 6 | ||
d (ℓ = 2) | 4dxy, 4dyz, 4dxz, 4dz2, 4dx2–y2 (mℓ = −2, −1, 0, 1, 2) | 10 | ||
f (ℓ = 3) | 7 4f orbitals (mℓ = -3 to 3) | 14 |
Orbitals and corresponding quantum numbers up to n = 4
The first four subshells of a quantum shell are labelled s, p, d, and f (after the first letters of early terms used to describe spectral lines, ‘sharp’, ‘principal’, ‘diffuse’ and ‘fundamental’). Subsequent subshells present in the 5th quantum shell onwards are labelled g, h, i, … in alphabetical order.
These letters are also used to describe the type and shape of orbitals present in the subshell.
The s subshell is present from the 1st quantum shell onwards, and corresponds to ℓ = 0. Only 1 s orbital is present in each quantum shell, and it has a spherical shape which increases in size with increasing principal quantum number.
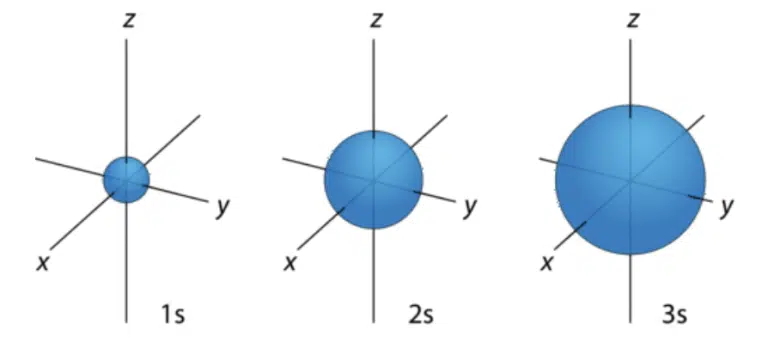
For ‘A’-Level Chemistry, representing s orbitals as a sphere with the x, y and z axes labelled is sufficient.
It may appear that the 2s orbital overlaps with the 1s orbital due to its size, but this is actually not true – s orbitals with principal quantum number 2 and above actually have spherical nodes inside which represent areas where there is zero probability of finding that particular electron.
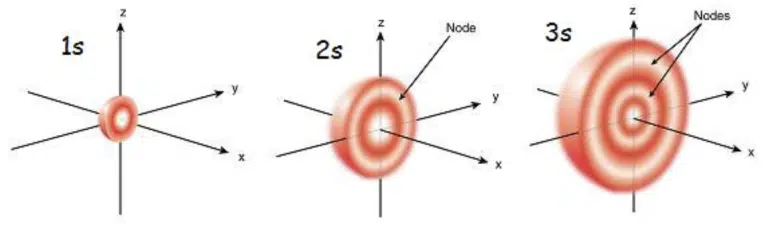
Cross section of s orbitals
p subshells are present from the 2nd quantum shell onward, and correspond to ℓ = 1. Each p subshell has 3 p orbitals, as there are three possible mℓ values: −1, 0, 1. Each p orbital is dumb-bell shaped, and differs by its orientation along the x, y and z axes.
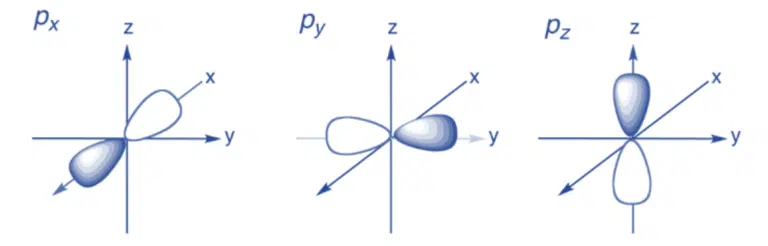
Within the same subshell, the three p orbitals all have the same energy level – we say that they are degenerate. Similar to s orbitals, the size of p orbitals increases with increasing principal quantum number.
The d subshell, which corresponds to ℓ = 2, is present from the 3rd quantum shell onwards, with each subshell containing 5 degenerate orbitals of identical energy.
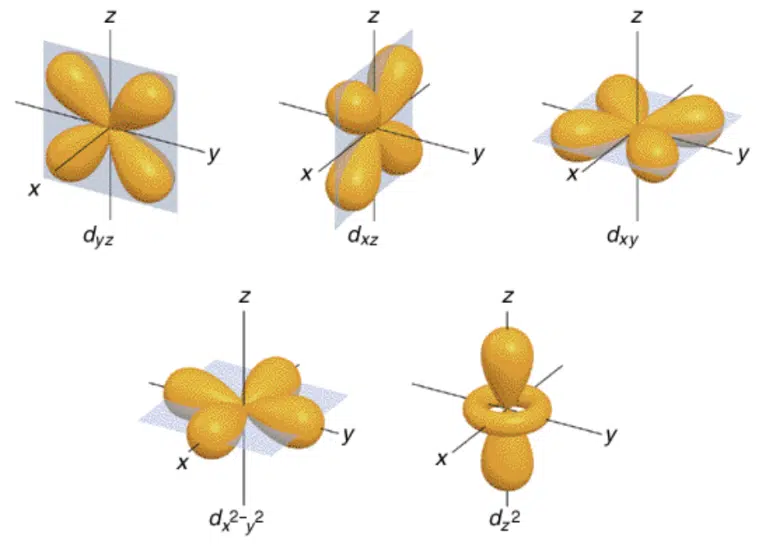
It is important to note that while all d orbitals in the same subshell have the same energy level, they do not all have the same shape or orientation.
The lobes of the dxy, dyz and dxz orbitals are between the axes of their respective planes, while the lobes of the dx2–y2 orbitals are arranged along the x and y axis. The dz2 orbital has lobes along the z axis only, with a ‘donut-shaped’ ring across the centre.
The positions of the lobes along or between the axes do not affect the energy levels in an isolated atom, but as you will learn in Transition Metals, may produce a split in energy levels when surrounded by negative charge in a ligand field.
The f subshell corresponds to ℓ = 3, and contains 7 orbitals as a total of 7 mℓ values are possible: −3, −2, −1, 0, 1, 2 and 3. They are present from the 4th principal quantum shell onward.
The shape of f orbitals is quite complicated – 4f orbitals can contain up to 8 lobes, and both the complexity and size increase with increasing principal quantum number. As such, drawing and identifying the f orbitals is not required nor expected for ‘A’-Levels, and are only included here for illustrative purposes.
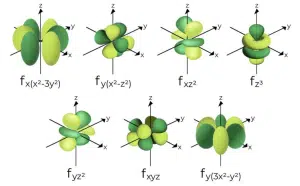
Shapes and orientations of 4f orbitals
Under the Bohr model, the electronic structure of atoms and ions is displayed in terms of which primary quantum shell they occupy; in the quantum model, the electronic structure is expanded to include the subshells they occupy.
Electronic Structure (Bohr Model) | Electronic Structure (Quantum Model) | Electron-in-box Diagram |
2.8.5 | 1s22s22p63s23p3 |
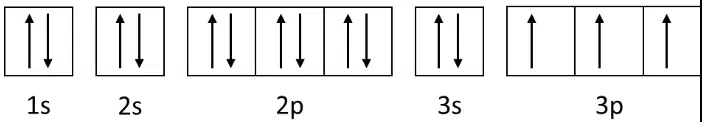
Representing the arrangement of electrons in a phosphorus (Z=15) atom
When writing the electronic structure of an atom or ion, it is important to keep in mind the following guidelines:
In the ground state of an atom or ion, electrons fill up subshells in order of increasing energy level.
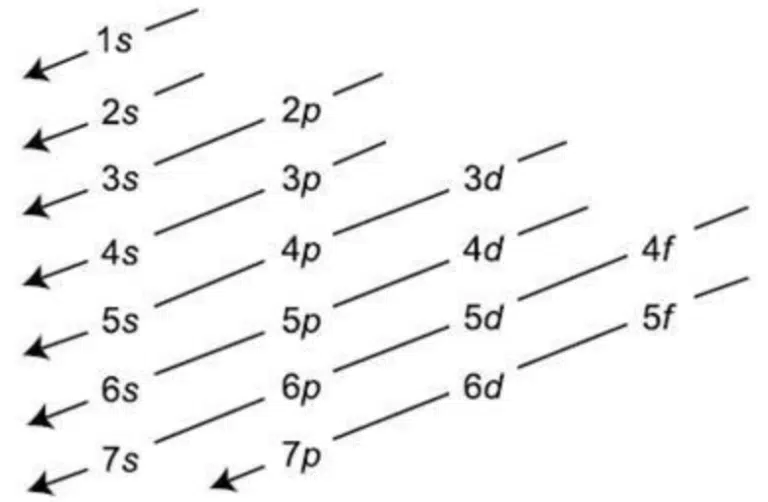
A common way of remembering the order of filling atomic subshells
When filling orbitals of equal energy (degenerate orbitals), each orbital must first be occupied singly with electrons of the same spin, before any of them can be paired with a second electron.
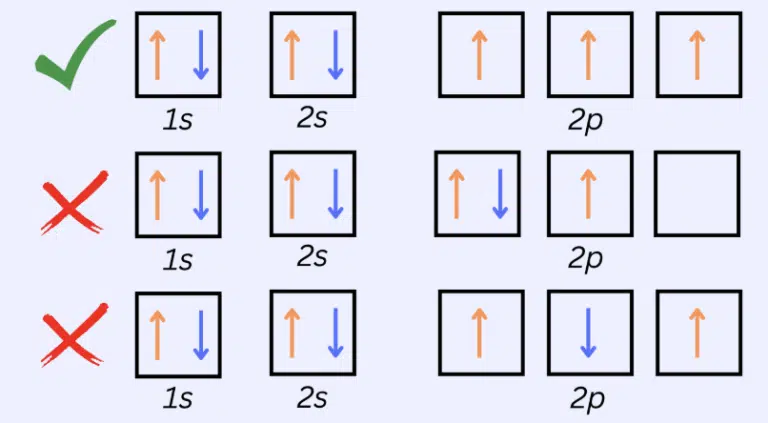
Since no two electrons can have the same set of quantum numbers, a pair of electrons occupying the same atomic orbital must have opposite spins.
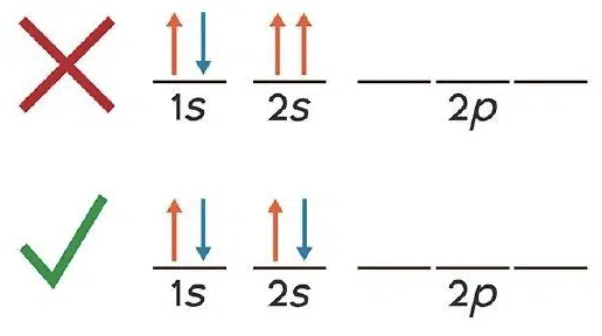
After having the Bohr model of atoms drilled in throughout secondary school, and even relying on it to explain important concepts like chemical bonding and reactivity, getting used to the quantum model of atoms may feel like one of the first big hurdles to overcome when starting ‘A’-Level Chemistry. Despite this, having an appreciation of atomic subshells and orbitals will definitely pay off in helping to explain some of the more complex ideas about bonding, molecular shapes, and energy which we will encounter later on.
Find this helpful? Find out more by joining us at Science of Studying!
Prepared by: Nadine
A little more about ourselves…
The Science of Studying provides live online tuition via Zoom classes for Combined Chemistry, Combined Biology, Pure Chemistry, Pure Biology, J1 Chemistry and Biology. To date, we have taught more than 800 students over 12 years.
In case you are wondering, yes – there is a science behind studying! At Science of Studying, we use our SOS systemTM to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind- numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
The SOS systemTM guides students through an effective process of:
– Understanding key concepts
– Applying the concepts through smart, targeted practice.
– Learning to avoid common ‘traps’ set by examiners
– Learning exam-smart answering techniques for each topic – Overcoming tricky exam questions
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry and Biology subjects, do reach out to us and we will see what we can do to help.
Or join our trial lessons to find our more at: https://thescienceofstudying.com/schedule/#trial
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)

The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us