- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM
Sometimes taking Chemistry in JC feels like walking into a Maths class by accident – we walk in expecting to learn about elements and compounds but get asked questions about angles, shapes and geometry instead. Not only do we need to learn the shapes of orbitals, we also need to deduce the shapes and bond angles of molecules just by looking at their formulae. How would we even describe the shapes of molecules when they are so small that we can’t observe them by the naked eye?
The answer is to use a model to predict how bonds will arrange themselves in a molecule, known as VSEPR Theory. It looks like some kind of knockoff brand or complicated programming language, but VSEPR stands for Valence Shell Electron Pair Repulsion. This basically sums up the key assumptions in this theory:
When electrons within the same atom repel each other, it destabilises the whole system, which means that the electron pairs will always try to arrange themselves in a way that minimizes the amount of repulsion between them. This is usually achieved by spacing themselves as far apart from each other as possible on the atom, which we will assume has a spherical shape.
But before you go on reading… You might want to download a pdf copy of this article as it is quite long!
Click the ‘Download’ button, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)

However, not all repulsion is equal: lone pairs and bond pairs repel each other to different extents. Lone pairs repel more strongly than bond pairs, which makes sense intuitively – bonding electrons are shared between two atoms, so the negative charge they produce is distributed over a wider area compared to lone pairs, which stay near only one atom.
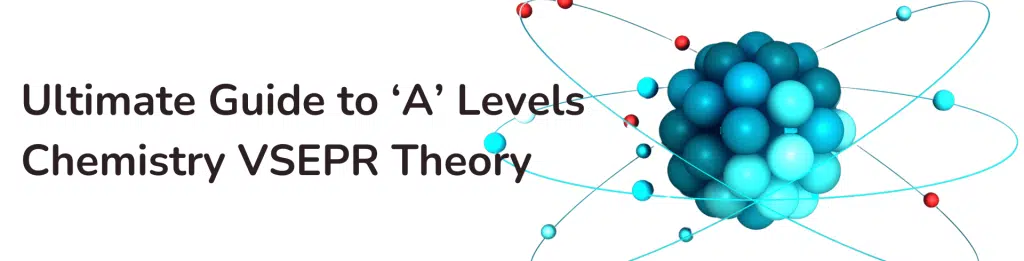
The central atom in a water molecule is O, and it has two bond pairs and two lone pairs
The order of repulsion strength is given by:
lone pair – lone pair > lone pair – bond pair > bond pair – bond pair
This means that when lone pairs are present in a molecule, they tend to “push” any bond pairs closer together.
What does any of this have to do with the shape of molecules or bond angles? Since the goal of electron pairs is to minimize the amount of repulsion between themselves, this means that they will tend to arrange themselves only in predictable geometries about the central atom that are mathematically proven to have the least amount of repulsion, and the angles between bonding pairs can be assumed to be in a fixed range based on these arrangements.
Therefore, using VSEPR theory, it is possible to accurately predict the shape of molecules just based on knowing the number of lone and bond pairs about the central atom, and even estimate the angles between covalent bonds.
It is important to remember that to use VSEPR Theory effectively, we must know how to draw dot-and-cross diagrams and/or Lewis diagrams of molecules in order to figure out how many lone pairs and bond pairs are present in the valence shell of the central atom. We must also take note that when considering molecular geometry, any covalent bond, whether single, double or triple, is treated as a “bond pair” even if there are more than two electrons involved, and any non-bonding unpaired electrons are treated as equivalent to a “lone pair” of two non-bonding electrons.
Suppose you had a molecule with two bond pairs on the central atom, for example, carbon dioxide (CO2). How would we predict the shape and bond angle of the molecule?

Recall that each double bond is treated as a single bond pair
Imagine the following situation: two people are about to be seated at a single circular table at a restaurant. They do not know each other, and would like to be seated as far apart from each other as possible in order to provide as much space as possible for each person. In this case, it becomes quite obvious that they would have to sit directly opposite each other, along the diameter of the circular table.
Seating arrangement about a circular table for two people
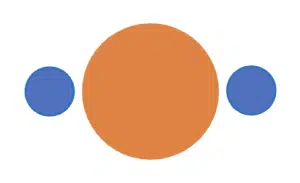
If we consider this problem in 3D space, with two objects positioned around a sphere instead, we also reach a similar conclusion – to maximise the space between the two objects, they must be situated on opposite ends of the sphere. If we were to measure the angle between the objects, it would be 180°.
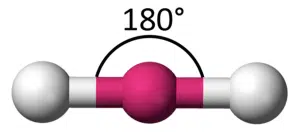
The sphere in the middle represents the central atom of the molecule, while the two objects represent the two atoms covalently bonded to the central atom. We say that the molecule has linear geometry, as all three atoms are connected in a straight line. The bond angle, which is measured between two covalent bonds, is 180°.
Now let’s extend the circular table scenario to seat three people. To maximise the space for each person, they would each have to be seated 120° apart from each other, at the vertices of an equilateral triangle:

Seating arrangement about a circular table for three people.
In 3D space, the solution looks similar to the 2D version – each object is positioned on the same plane at 120° around a sphere.
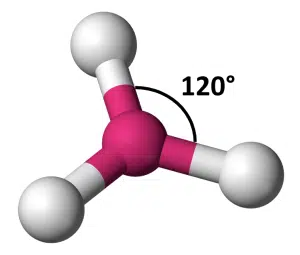
This is called trigonal planar geometry. The bond angle between any two of the three bonds is equal at 120°.
Now what happens if one of the bond pairs is replaced with a lone pair?
In the original 3 bond pair case, the bond angles are equal because the repulsion between each bond pair is equal. However, lone pair-bond pair repulsion is greater than bond pair-bond-pair repulsion, so when a lone pair is present, the bond angles between the remaining bond pairs will be affected.
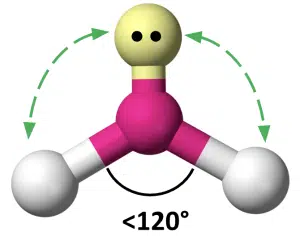
The lone pair repels the bond pairs more, reducing the bond angle below 120°
Only bond pairs are considered when determining the shape of the molecule, so while the electron pairs are still arranged in a trigonal planar geometry, the bond pairs are angled or in a V-shape arrangement. We say that the molecular shape is bent. The exact bond angle depends on other factors such as the strength of repulsion, length of bonds and relative electronegativity of the atoms, but will typically range between 115° to 119°.
Once we consider 4 objects arranged to minimize repulsion, the ideal arrangement in 2D and 3D becomes very different. While we can imagine that the best way to seat 4 people around the circular table would be placing them at right angles to each other, in 3D space, the best arrangement is what we call tetrahedral geometry. The 4 objects are arranged at the vertices of a tetrahedron, and the angle between any two of them is equal at 109.5°.
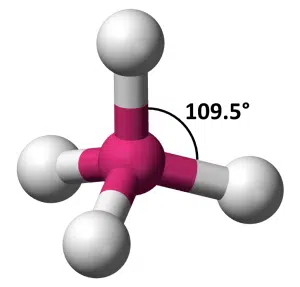
A tetrahedron is a 3D shape with 4 triangular faces, 6 straight edges and 4 vertices.

4-sided dice are regular tetrahedrons
This means that if we have a molecule with 4 bond pairs about the central atom, such as CH4, it will have a tetrahedral shape, with bond angles of 109.5° between any two C-H bonds.
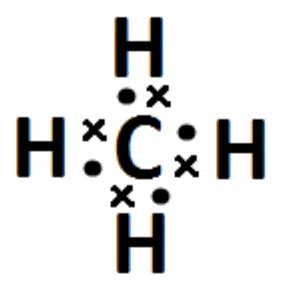
Dot and cross diagram of CH4
If one of the bond pairs was replaced with a lone pair, giving us 3 bond pairs and 1 lone pair, the electron arrangement would still be tetrahedral. However, since the lone pair repels the 3 bond pairs more strongly, they are pushed closer together, reducing the bond angle to between 106° to 107°. Since the three bonds form a pyramid shape, we call this geometry trigonal pyramidal geometry.
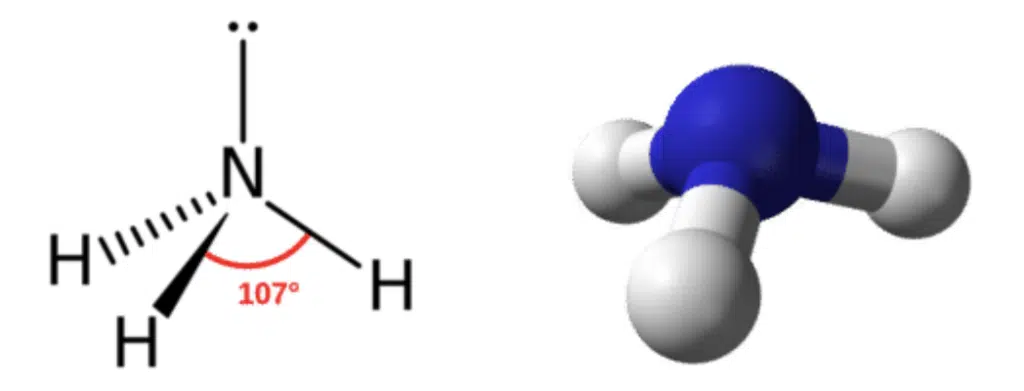
Ammonia is a good example of a molecule with trigonal pyramidal geometry
What happens when we change another bond pair to a lone pair?
Now we have 2 bond pairs and 2 lone pairs, which are arranged in tetrahedral geometry. However, since the strength of repulsion decreases in order of lone pair-lone pair > lone pair-bond pair > bond pair-bond pair, we expect the bond pairs to be pushed even closer together, because the two lone pairs repel each other very strongly, and in turn repel the bond pairs strongly as well.
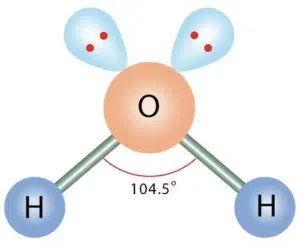
A classic example of a structure with two bond pairs and two lone pairs is water.
The shape of the molecule is bent, and the bond angle between the two remaining bonds is typically between 104° to 105°. For water specifically, the bond angle is 104.5°.
Hopefully these examples have helped you understand the general strategy we use to determine molecular geometry: first, determine the total number of electron pairs to decide on the basic electron pair geometry. Then, adjust the molecular shape based on the actual number of lone pairs and bond pairs, and determine the bond angle.
With 5 pairs of electrons, the optimal geometry no longer has equal angles between any two electron pairs. The basic geometry is called trigonal bipyramidal, as it looks like two triangular pyramids joined together.
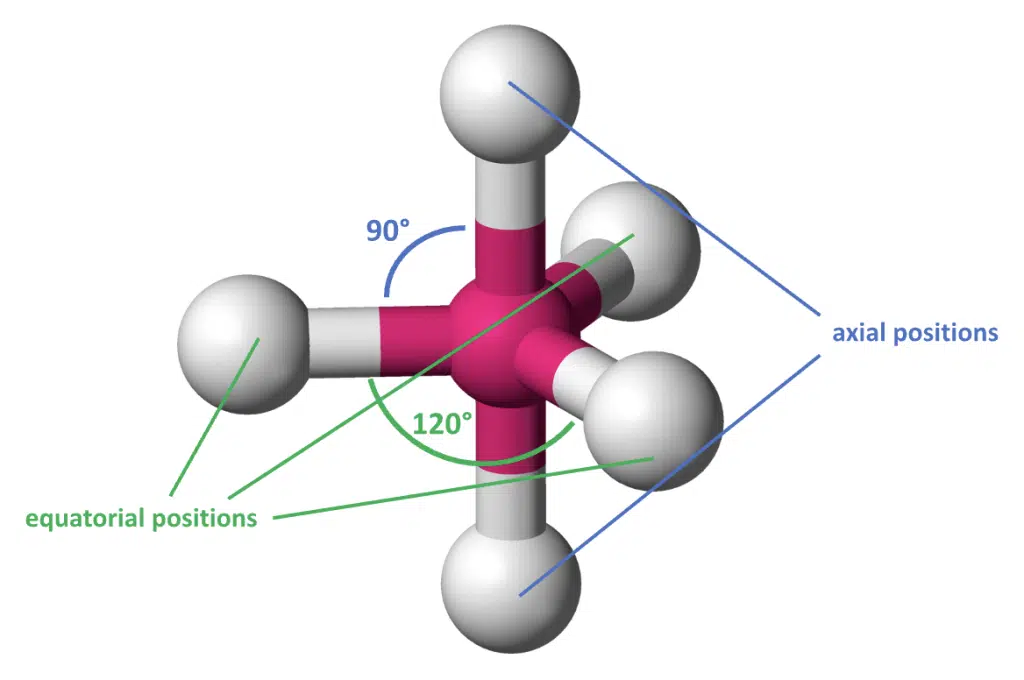
With 5 atoms bonded to the central atom (5 bond pairs), three of them are arranged along the same plane as the central atom at 120° angles in equatorial positions, while the two remaining atoms are above and below the plane at axial positions, forming 90° angles with the three equatorial atoms. Take note that both bond angles must be quoted when describing trigonal bipyramidal geometry.
When 1 bond pair is switched with a lone pair, we have 4 bond pairs and 1 lone pair. To minimize repulsion, the lone pair is placed in an equatorial position, producing a seesaw geometry.

With 5 atoms bonded to the central atom (5 bond pairs), three of them are arranged along the same plane as the central atom at 120° angles in equatorial positions, while the two remaining atoms are above and below the plane at axial positions, forming 90° angles with the three equatorial atoms. Take note that both bond angles must be quoted when describing trigonal bipyramidal geometry.
When 1 bond pair is switched with a lone pair, we have 4 bond pairs and 1 lone pair. To minimize repulsion, the lone pair is placed in an equatorial position, producing a seesaw geometry.
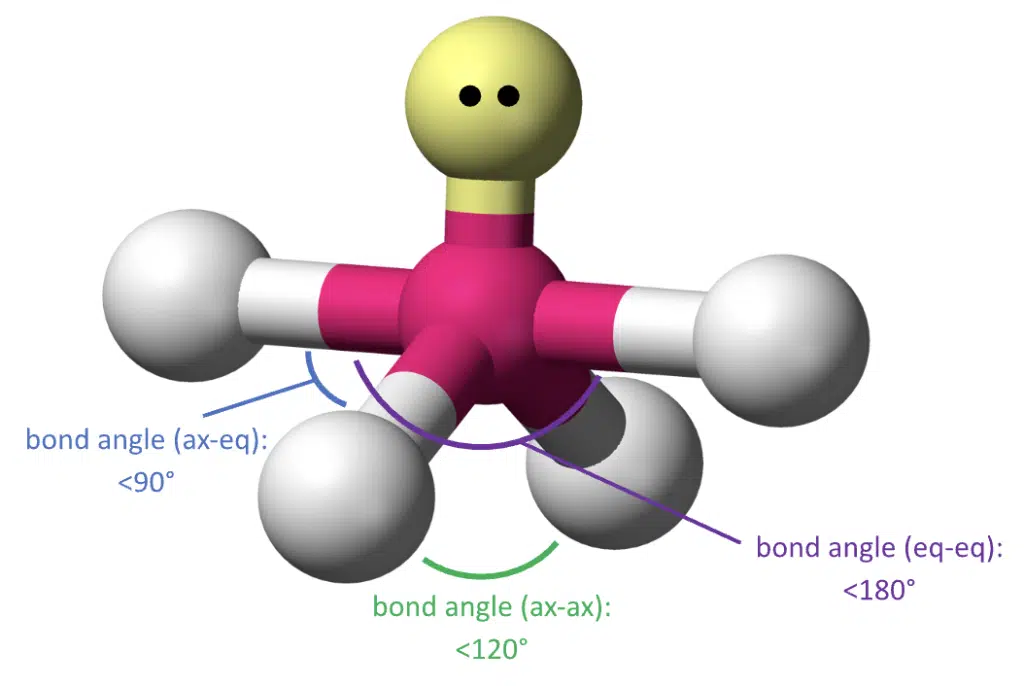
The increased repulsion from the lone pair also decreases the bond angles between axial and equatorial bonds.
With 3 bond pairs and 2 lone pairs, another equatorial bond is replaced with a lone pair, and the resulting molecular geometry is T-shaped.
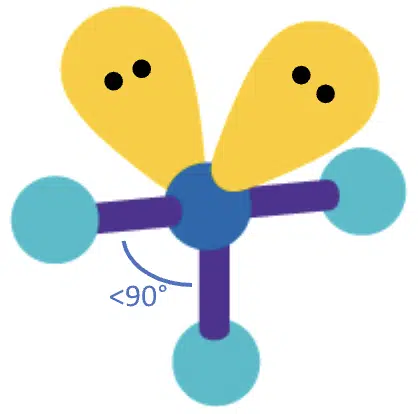
If 2 bond pairs and 3 lone pairs are present, then all the lone pairs take up the equatorial positions, so the molecular geometry becomes linear. Since all three lone pairs repel the bond pairs equally, the bond angle becomes 180°.
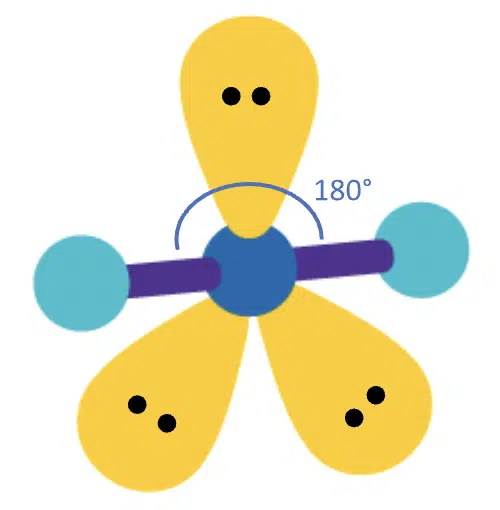
(Adapted from Sigma-Aldrich website)
With 6 bond pairs, the basic geometry is octahedral, and each bond angle is equal at 90°. You can imagine an octahedron as two square-base pyramids stuck together at the bases.
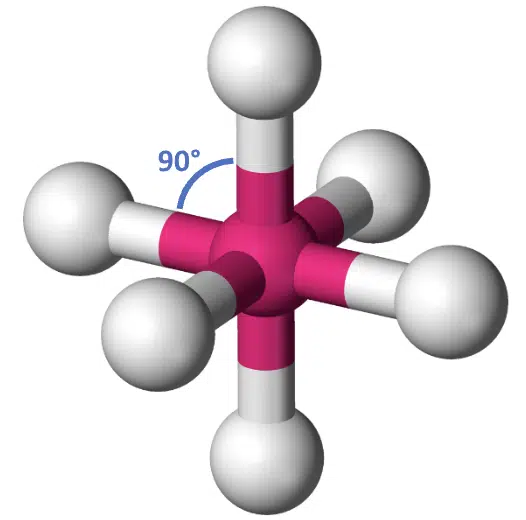
Octahedral molecular geometry

An 8-sided die also has an octahedral shape.
If one of the bond pairs is replaced with a lone pair, we would have 5 bond pairs and 1 lone pair. The resulting molecular geometry is square pyramidal, with bond angles smaller than 90°.
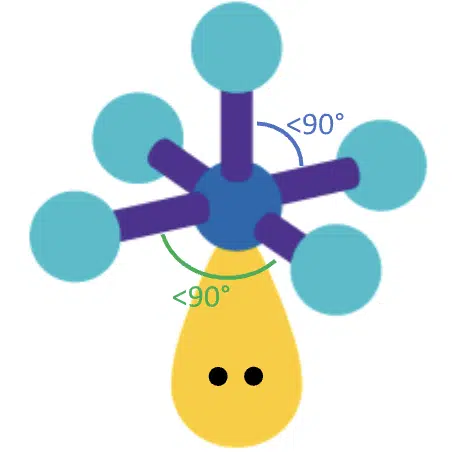
(Adapted from Sigma-Aldrich website)
With 4 bond pairs and 2 lone pairs, the lone pairs will take up opposite positions, so the resulting molecular geometry is square planar with bond angles of 90°.
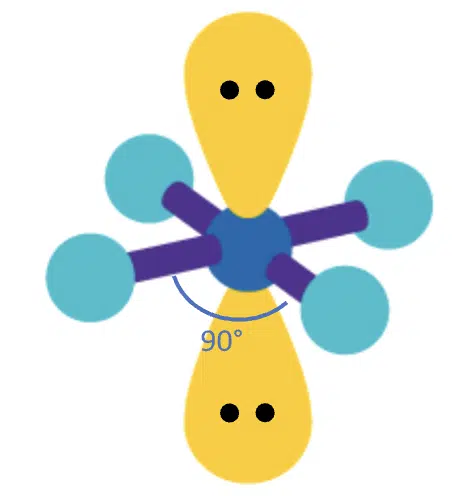
(Adapted from Sigma-Aldrich website)
3 bond pairs and 3 lone pairs produces a T-shape molecular geometry (comparable to 3 bond pairs and 2 lone pairs, shown in the previous section). Similarly, 2 bond pairs and 4 lone pairs produces a linear molecular geometry (comparable to 2 bond pairs and 3 lone pairs).
The number of lone pairs and bond pairs and corresponding geometries are summarized in the table below:
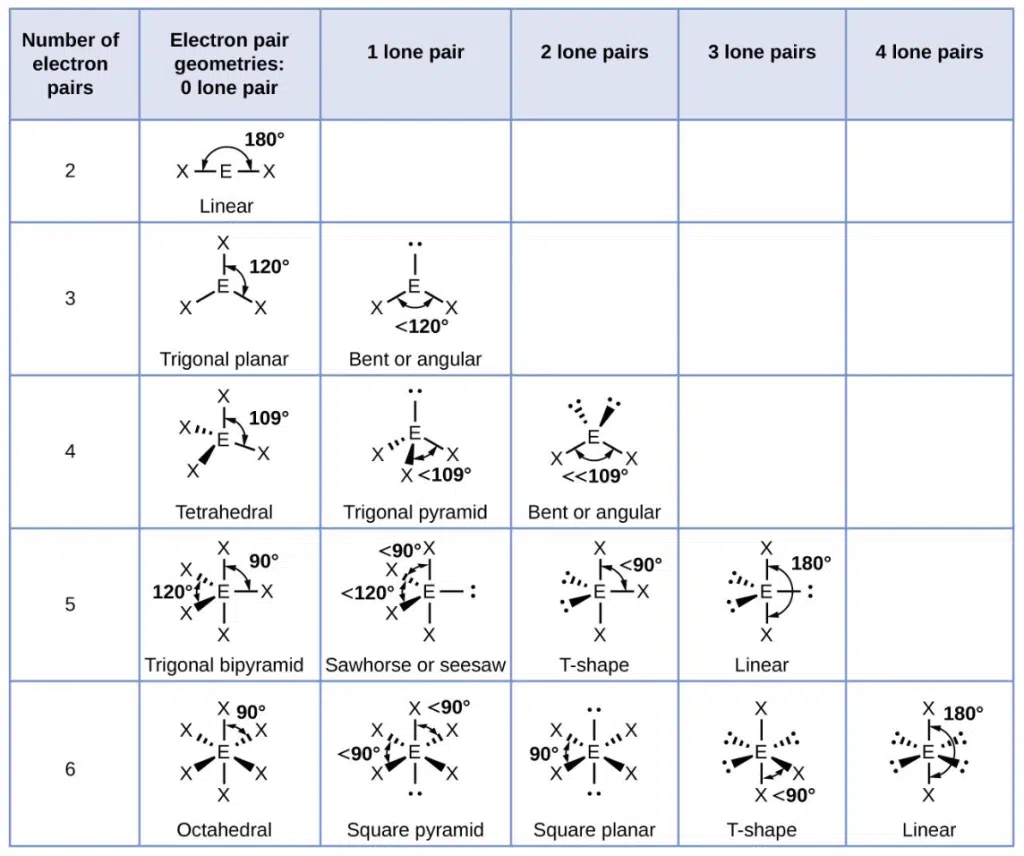
It is important to take note that it is possible for two molecules to have different bond angles even when they have the exact same number of lone pair and bond pairs, as the exact bond angle may be affected by the lengths of the bonds, and relative electronegativity of the central and side atoms.
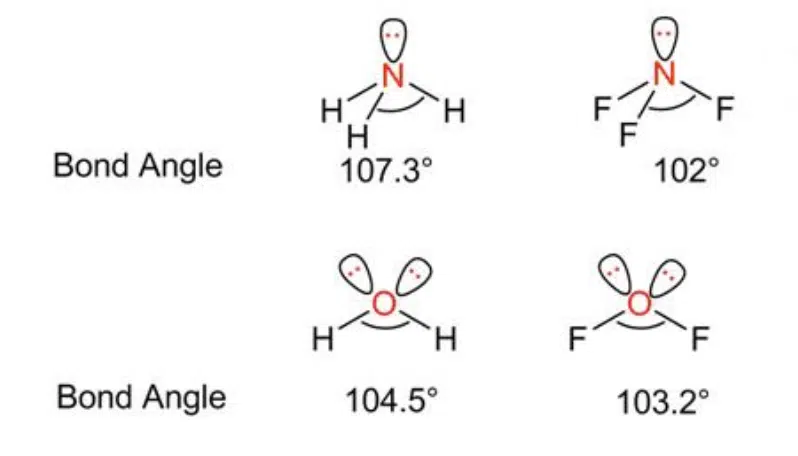
Examples of molecules with the same geometry but different bond angles.
In both cases illustrated above, fluorine is more electronegative than hydrogen, so the bonding pair of electrons is more strongly attracted to fluorine than the central atom. This results in each bond producing less electron density about the central atom, so there is less repulsion between the bond pairs. As a result, the bond pairs are pushed closer together and a smaller bond angle is achieved.
One more thing to note is that the same name may be used to describe molecular geometries with fundamentally different numbers of lone pairs and bond pairs. For example, molecules with 1 lone pair and 2 bond pairs, and 2 lone pairs and 2 bond pairs are both described as bent in shape, but have different bond angles due to their different basic electron pair geometry.
With this, you are now a VSEPR Master, and you will be able to tell the geometry and bond angles of molecules by looking at their formulae – just remember to draw the dot-and-cross diagrams and count the number of lone and bond pairs to be sure!
Find this helpful? Find out more by joining us at Science of Studying!
Or join our trial lessons to find our more at: https://thescienceofstudying.com/schedule/#trial
Prepared by: Nadine
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us