- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

The Data Booklet is your best buddy during your Chemistry exams. With the right approach, it can significantly enhance your ability to solve problems efficiently and accurately. To master the use of the Data Booklet, let’s go through what exactly is inside the Data Booklet and how you can use it effectively during exams.
But before you go on reading… You might want to download a pdf copy of this article as it is quite long!
Click the ‘Download’ button, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)

S/N | Content | Description |
1 | Important values, constants and standards | This section includes constants like the gas constant, Avogadro’s number, and molar volume of gas. These values are crucial for calculations across various topics. |
2 | Ionisation energies (1st, 2nd, 3rd and 4th) of selected elements in kJ mol–1 | This section includes all ionisation energies of selected elements. You should be able to use these values to explain the factors influencing the ionisation energies of element, which is covered in ‘Atomic Structure’ and ‘Periodic Table’. |
3 | Bond energies | This section includes bond energies of (a) diatomic and (b) polyatomic molecules. These values are important for your calculations of enthalphy changes from bond energies in ‘Chemical Energetics’. |
4 | Standard electrode potential and redox potentials, E⦵ at 298 K (25°C) | This section includes the standard electrode (redox) potentials, E⦵, in (a) alphabetical order or (b) decreasing order of oxidising power (i.e., most positive E⦵ to most negative E⦵). These values can be useful to predict spontaneity of redox reactions, determine standard cell potential, E⦵cell, and apply the relationship ΔG⦵ = –nFE⦵. This will be covered in ‘Electrochemistry’. |
5 | Atomic and ionic radii | This section includes the atomic and ionic radii of selected element. You can expect to use these values when discussing about atomic and ionic radii in ‘Atomic Structure’ and ‘Periodic Table’. |
6 | Typical proton (1H) chemical shift values (δ) relative to TMS = 0 | Not applicable for H2 Chemistry (although some advanced questions may require you to use information from this section) |
7 | Characteristic infra-red absorption frequencies for some selected bonds | Not applicable for H2 Chemistry (although some advanced questions may require you to use information from this section) |
8 | The orientating effect of groups in aromatic substitution reactions | Not applicable for H2 Chemistry (although some advanced questions may require you to use information from this section) |
9 | Qualitative Analysis Notes | This section includes the tests and observations for (a) aqueous cations, (b) anions, and (c) gases, as well as the (d) colour of halogens. This section is extremely useful for qualitative analysis questions and can help you solve questions without needing to recall everything from memory. |
10 | The Periodic Table of Elements | The periodic table in the data booklet provides atomic numbers, relative atomic masses, and other essential properties. It’s your go-to resource for questions involving ‘Atomic Structure’, ‘Stoichiometry’, and ‘Periodic Table’. |
Sample Question – Catholic Junior College 2017 JC1 Promos Section A Q8
In a pathology laboratory, a sample of urine containing 0.120 g of urea, NH2CONH2, (Mr = 60.0) was treated with an excess of nitric(III) acid. The urea reacted according to the following equation. NH2CONH2(aq) + 2HNO2(aq) → CO2(g) + 2N2(g) + 3H2O(l) The gas produced was passed through aqueous potassium hydroxide and the final gas collected. How many molecules of the final gas were collected at room temperature and pressure? A 2.41 x 1020 B 1.20 x 1021 C 2.41 x 1021 D 3.61 x 1021 |
Answer: C
Amount of urea, NH2CONH2 = mass/Mr = 0.120/60.0 = 0.00200 mol CO2(g), which is an acidic gas, was removed by the base, KOH(aq). Hence, N2(g), which is a neutral gas, was the final gas collected. NH2CONH2 2N2 Amount of N2 = 2 x 0.00200 = 0.00400 mol Number of N2 molecules = 0.00400 x 6.02 x 1023 (Avogadro’s constant taken from Data Booklet) = 2.41 x 1021 |
Sample Question – Victoria Junior College 2017 JC1 Promos Paper 1 Section A QS2(b)
(i) State the electronic configuration of phosphorous and sulfur. (ii) With reference to the Data Booklet, explain how the first ionisation energy of phosphorus is compared to that of silicon and sulfur. |
Answer:
(i) P: 1s2 2s2 2p6 3s2 3p3 S: 1s2 2s2 2p6 3s2 3p4 (ii) The first ionisation energies of phosphorus, silicon and sulfur are 1060, 786 and 1000 kJ mol-1 respectively (taken from Data Booklet). The first ionisation energy of phosphorus is higher than that of silicon because it has one more proton thus greater nuclear charge but similar shielding effect due to same number of inner shell electrons, thus greater effective nuclear charge. The first ionisation energy of phosphorus is higher than sulfur as the electrons removed from sulphur is from a doubly occupied orbital while that of phosphorus is from a singly occupied orbital. Thus, the inter-electronic repulsion between electrons in the same orbital leads to a lower first ionisation energy. 3. Bond EnergiesSample Question – Anglo-Chinese Junior College 2017 JC1 Promos Section D Q1(a)
Answer: |
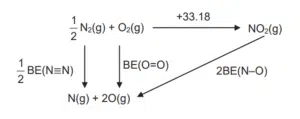
33.18 = ½ x BE(NN) + BE(O=O) – 2 x BE(N-O)
Substituting bond energies values from Data Booklet, 33.18 = ½ x 944 + 496 – 2 x BE(N-O) BE(N-O) = +467 kJ mol-1 |
Sample Question – Jurong Junior College 2017 JC1 Promos Paper 2 Section A Q5(a)
The diagram shows the apparatus used to measure the standard cell potential, E⦵cell, when Fe3+(aq)/Fe2+(aq) half-cell is connected to Cu2+(aq)/Cu(s) half-cell.
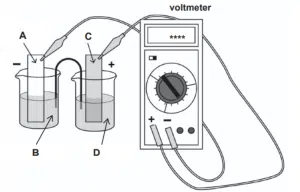
(i) Identify the substance(s) represented by the letters A to D. Specify the required conditions where necessary.
(ii) Determine the value of the standard cell potential, E⦵cell, that will appear on the display of the voltmeter.
Answer:
(i) A Cu(s) B Cu2+(aq), 1 mol dm-3 C Pt(s) D Fe2+(aq), 1 mol dm-3 AND Fe3+(aq), 1 mol dm-3 (ii) Taking standard electrode potential values from Data Booklet, E(Fe3+/Fe2+) = +0.77 V E(Cu2+/Cu) = +0.34 V E⦵cell = +0.77 – (+0.34) = +0.43 V |
Sample Question – Jurong Junior College 2017 JC1 Promos Section C Q1(b)
With reference to relevant values in the Data Booklet, explain how the lattice energy of Fe2O3 would compare to that of Al2O3. |
Answer:
Lattice energy is proportional to (q+q–)/(r++r–). While Fe3+ and Al3+ have the same charge, Fe3+ has a larger ionic radius of 0.055 nm compared to that of Al3+ at 0.050 nm (taken from Data Booklet). Fe2O3 would have a less exothermic lattice energy as compared to that of Al2O3. |
Sample Question – Hwa Chong Institution 2017 JC1 Promos Paper 1 Q19
Which reaction sequence will produce the best yield of 2-bromo-4-nitrobenzoic acid from methylbenzene?
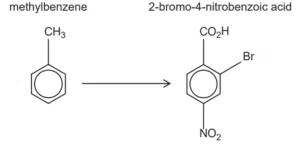
A bromination → oxidation → nitration
B nitration → bromination → oxidation
C nitration → oxidation → bromination
D oxidation → nitration → bromination
Answer:
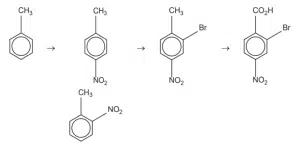
This question is about the orientating effect of groups in electrophilic substitution reactions. Use Data Booklet Section 8 to find the best sequence that gives the fewest number of products other than the desired one (i.e. best yield of desired product). Sequence B is the best choice.
To master the use of the Data Booklet, regularly flip through the booklet to familiarise yourself with where specific information is located. This will save precious time during exams. Remember to incorporate the booklet into your daily practice by using it while tackling practice questions, which will help reinforce familiarity and improve time management.
Find this helpful? Find out more by joining us at The Science of Studying!
Prepared by: Tan Wee Leng
This article is written based on the 9729 Chemistry GCE Advanced Level H2 Syllabus. https://www.seab.gov.sg/docs/default-source/national-examinations/syllabus/alevel/2024syllabus/9729_y24_sy.pdf
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us