- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

Before you read on, you might want to download this entire revision notes in PDF format to print it out for your child, or yourself to read it later.
This will be delivered to your email inbox.
Encountering organic chemistry for the first time might feel like suddenly being asked to speak a completely different language. Within the first few lessons, students are overloaded with unfamiliar words like ‘hydrocarbons’, ‘homologous series’, ‘functional group’ among many others, and expected to learn their definitions and use them accurately.
To make matters more difficult, teachers and students often try to blitz through the organic chemistry chapters at breakneck speed, because it is the Common Last Topic that stands in the way between normal curriculum time and full exam preparation, when having an extra one or two weeks available can make a huge difference.
With so many new and similar-sounding words thrown at them in rapid-fire succession, and the added pressure to remember them quickly so that you can have more time to focus on your prelims and ‘O’-Levels, it is only natural that many students may get lost and confused along the way. However, these small misconceptions can not only cost your grade during exams, but could lead to even bigger misunderstandings and confusion if you decide to take a biology or chemistry related course in the future, where organic chemistry becomes even more relevant and complicated.
Ironing out misconceptions early is the best way to build a solid foundation in organic chemistry. To help you out, in this post, we’ll talk about the key families of organic compounds, how to identify them, and some of their important reactions and uses.
A functional group is an atom or group of atoms that gives an molecule its distinct chemical properties. Different molecules with the same functional group will undergo the same reactions characteristic to that functional group, regardless of any other functional groups or atoms they may have; they have the same chemical properties.
There are countless organic molecules found in nature, and they wildly vary in terms of size, structure and bonding; some are made up of a small handful of atoms, while others have complex chains of tens of thousands of atoms. Hence, instead of studying the properties and reactions of each compound individually, organic chemists find it much more useful to classify molecules that have the same functional group together, and focus on the unique reactions of each functional group.
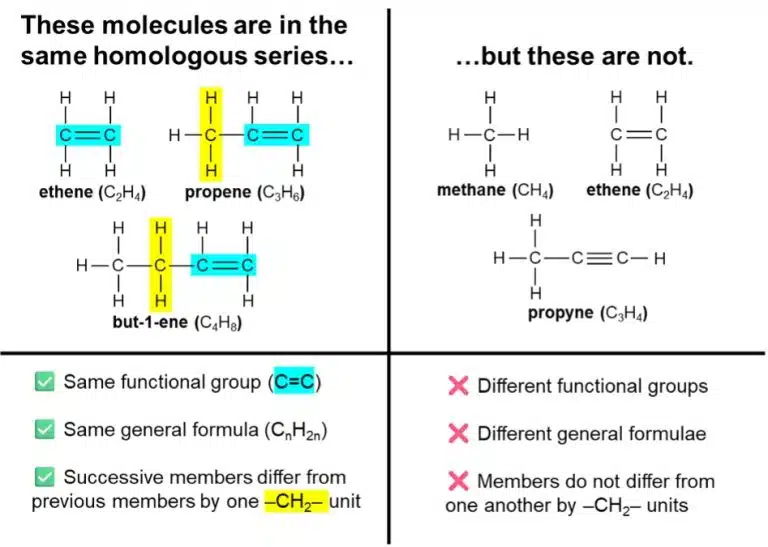
A homologous series is a family of organic molecules that meet the following requirements:
Compounds in the same homologous series are given similar names, and undergo the same chemical reactions.
A functional group refers to a particular atom or group of atoms on a molecule that is responsible for the chemical reactions it undergoes – in other words, it gives the molecule its chemical properties.
A homologous series is a group of molecules that are similar in specific ways, which include having the same functional group and the same general formula. By extending the longest carbon chain of a molecule by one or more –CH2– units, we can deduce other members in its homologous series.
In ‘O’-Level Chemistry, we usually study organic molecules in terms of homologous series and their characteristic functional groups; however, do take note that two molecules just having the same functional group may not necessarily put them in the same homologous series.
The table below shows the four main homologous series tested in ‘O’-Level Chemistry, their general formulae and functional groups:
Homologous Series | Functional Group | Structure of Functional Group | General Formula |
alkanes | (none) | – | CnH2n+2 (n = 1, 2, 3, …) |
alkenes | carbon-carbon double bond | C=C | CnH2n (n = 1, 2, 3, …) |
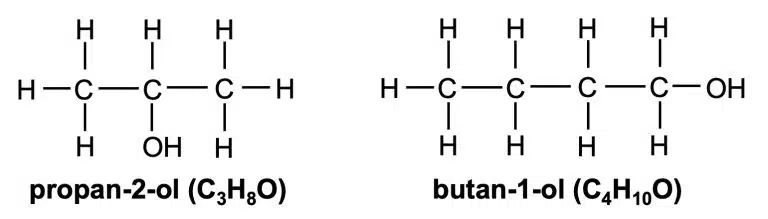
alcohols | hydroxyl | –OH | CnH2n+1OH (n = 1, 2, 3, …) |
carboxylic acids | carboxyl | –COOH | CnH2n+1COOH (n = 0, 1, 2, …) |
Alkanes are a class of organic molecules that are made up of only carbon and hydrogen atoms – we call such compounds hydrocarbons. What sets alkanes apart from other hydrocarbons is that they only contain carbon-carbon (C–C) and carbon-hydrogen (C–H) single bonds; they do not have a unique functional group. We say that alkanes are saturated hydrocarbons as they only contain single bonds.
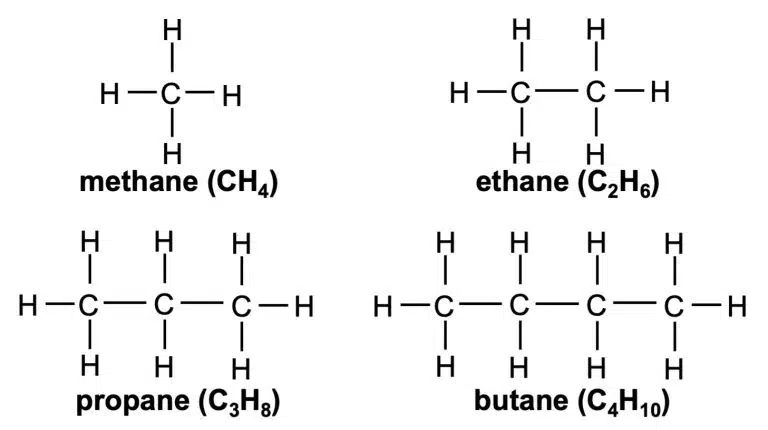
The general formula of alkanes is CnH2n+2 where n starts at 1. So, an alkane with 5 carbons will have the formula C5H2(5)+2 = C5H12.
It is important to take note of the trends in physical properties of alkanes as the carbon chain gets longer (the number of carbons increases). With increasing carbon chain length:
Alkanes have many uses, including as solvents, fuel and as starting materials for more complex chemical products. The main source of alkanes is from crude oil, which is separated or refined into different fractions through fractional distillation.

However, compared to other homologous series, alkanes are generally unreactive. The main reactions of alkanes are combustion in oxygen and substitution with halogens.
Alkanes burn in excess oxygen to form carbon dioxide and water only. This process is called complete combustion.
C3H8 (g) + O2 (g) → 3 CO2 (g) + 4 H2O (l)
However, if limited oxygen is available, the alkane undergoes incomplete combustion, forming products such as carbon monoxide or unburnt carbon instead. For example,
2 C3H8 (g) + 7 O2 (g) → 6 CO (g) + 8 H2O (l)
C3H8 (g) + O2 (g) → 3 CO2 (g) + 4 H2O (l)
A substitution reaction is one in which an atom or group of atoms in a molecule is replaced by another atom or group of atoms.
Alkanes can undergo substitution reactions with chlorine or bromine in the presence of UV light. It is considered a photochemical reaction because UV light is required to start the reaction.
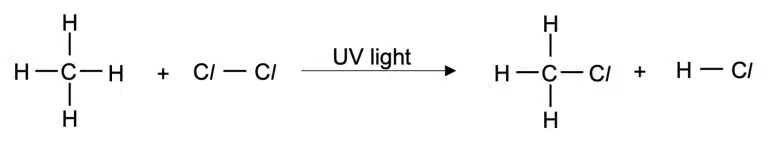
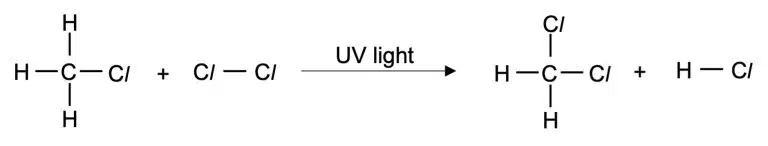
If the reaction is carried out with excess halogen, multiple substitutions can occur.
Alkenes are hydrocarbons like alkanes, but they are unsaturated as they have carbon-carbon double bonds (C=C) as their functional group. This makes them more reactive and able to undergo more types of reactions at milder conditions compared to alkanes.
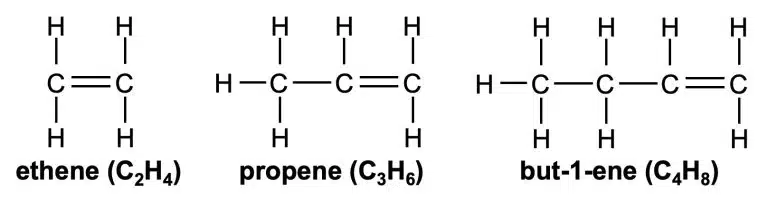
The general formula of alkenes is CnH2n; an alkene will always have 2 less hydrogens compared to an alkane with the same number of carbons. For example, the formula of the alkene with 5 carbons is C5H10.
Alkenes can be produced from alkanes by cracking, which is a process in which longer-chain hydrocarbons are broken down into shorter-chain hydrocarbons and/or hydrogen.
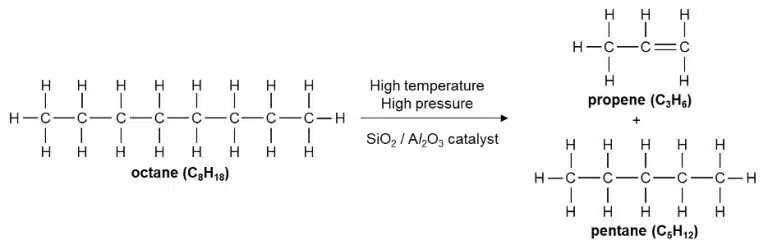
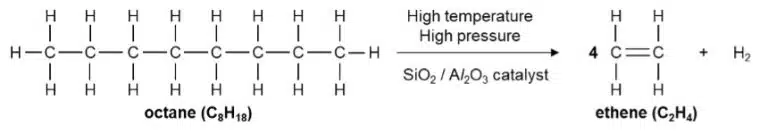
Like alkanes, alkenes also undergo combustion in oxygen, and exhibit similar changes in physical properties as the length of their carbon chain increases. Unlike alkanes, alkenes undergo addition reactions instead of substitution reactions with halogens.
Alkenes can be produced from alkanes by cracking, which is a process in which longer-chain hydrocarbons are broken down into shorter-chain hydrocarbons and/or hydrogen.
An addition reaction is one in which two or more molecules combine to make a larger molecule. Only one product (the larger molecule) is obtained, and there are no side products.
The unsaturated C=C bond on alkenes allows them to undergo addition reactions with several different reactants, most notably halogens such as chlorine and bromine.

Addition reactions are useful in telling alkanes and alkenes apart; the distinguishing test for alkenes (and other compounds containing C=C bonds) is to add bromine water to the sample. If the sample contains an alkene, it will react with the bromine and decolourise the yellow-brown solution, turning it colourless. However, if the sample does not contain alkenes or any unsaturated compound, the solution remains yellow-brown.

Alkenes also undergo addition reactions with hydrogen gas (hydrogenation) to form alkanes, and steam to form alcohols (hydration). A large number of alkene molecules can also join together to form a very long molecule known as a polymer, in a process called addition polymerisation.

Alcohols are organic molecules that contain the hydroxyl (–OH) functional group. Compared to alkanes and alkenes of similar molecular mass, alcohols tend to be more soluble in water, and have higher melting and boiling points. They have the general formula CnH2n+1OH.
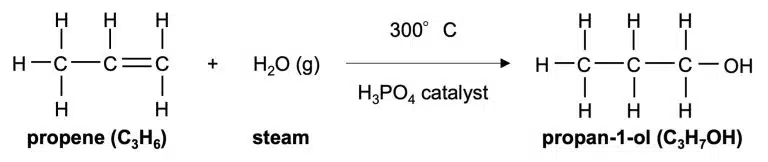
Alcohols are produced by one of two ways; by addition reactions between steam and alkenes, or by fermentation from sugars in the case of ethanol specifically.
Micro-organisms such as yeast can convert sugars into ethanol in anaerobic conditions. This process is called fermentation.
2 C6H12O6 (aq) → 2 C2H5OH (g) + 2 CO2 (l)
Yeast is sensitive to temperature changes; they work best around 37°C and will denature if the temperature is too high. It is also important to note that the fermentation process will stop when the alcohol content reaches about 15%, because high concentrations of alcohol will kill the yeast as well.
Ethanol is found in alcoholic drinks, and is also a good solvent and disinfectant. It is also used as a biofuel, as it can be burnt to produce heat energy and can be produced cheaply from plant matter.

Like most organic molecules, alcohols can undergo combustion to produce carbon dioxide and water.
C2H5OH (l) + O2 (g) → 2CO2 (g) + 3H2O (l)
Alcohols can also be oxidised into carboxylic acids by oxidising agents like acidified potassium manganate(VII), or by oxygen in the presence of bacteria.
Alcohols are converted to carboxylic acids when they undergo oxidation.
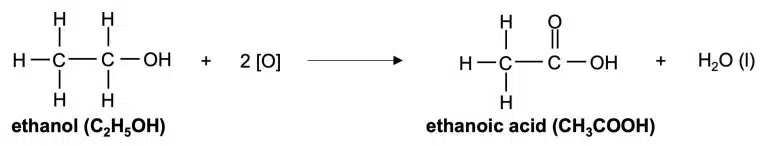
In the above equation, [O] represents oxygen from an oxidising agent.
When heated with acidified potassium manganate(VII), alcohols will decolourise the purple solution.
Alcohols can also be oxidised by atmospheric oxygen in the presence of bacteria. This is why wine and other alcoholic drinks can go sour after being left exposed to air for a long period of time.
C2H5OH (aq) + O2 (g) → 2CH3COOH (aq) + H2O (l)
Carboxylic acids are organic compounds containing the carboxyl (–COOH) functional group. As their name suggests, they are weak acids because they partially dissociate in water to produce hydrogen (H+) ions.
They have the general formula CnHn+1COOH. Take note that since the carbon on –COOH counts as part of the carbon chain, n starts at 0 instead of 1.
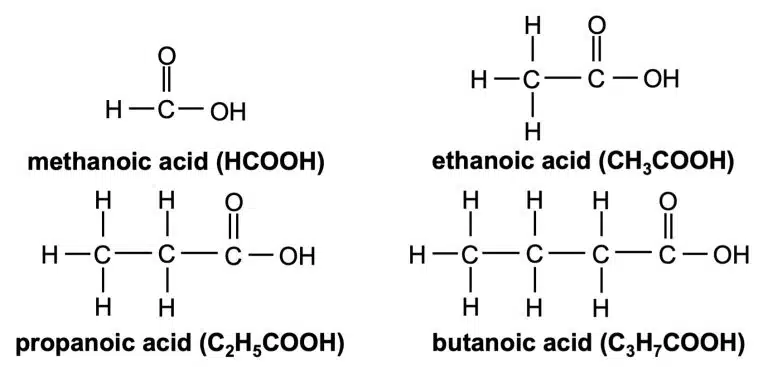
Carboxylic acids share the same general physical properties as inorganic acids:

Carboxylic acids also undergo the same reactions as acids in general:
They react with bases to form salts and water
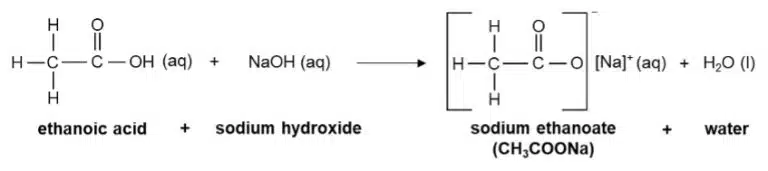
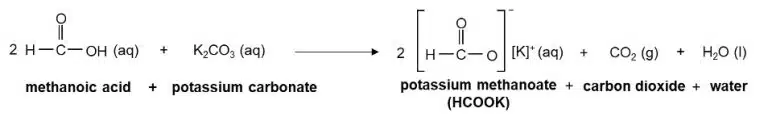
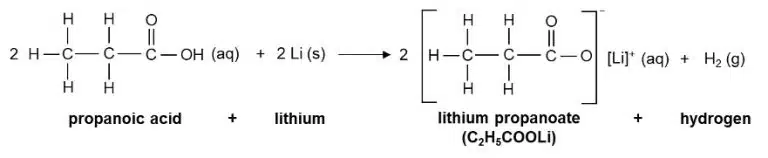
The salts of carboxylic acids are called carboxylate salts, and their ions follow similar naming conventions to their parent acid. For example, methanoic acid (HCOOH) forms the methanoate ion (HCOO−), and ethanoic acid (forms the ethanoate ion (CH3COO−).
Carboxylic acids can also react with alcohols to form esters in a process called esterification.
Esters are a class of organic molecule that contain the ester (–COO–) linkage. They are produced when alcohols react with acids in a condensation reaction known as esterification.
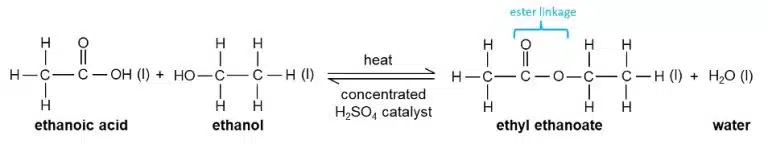
A condensation reaction is one in which two or more molecules react to form a larger molecule, giving out a small molecule such as water in the process. This sets it apart from addition reactions, where only one product is formed.
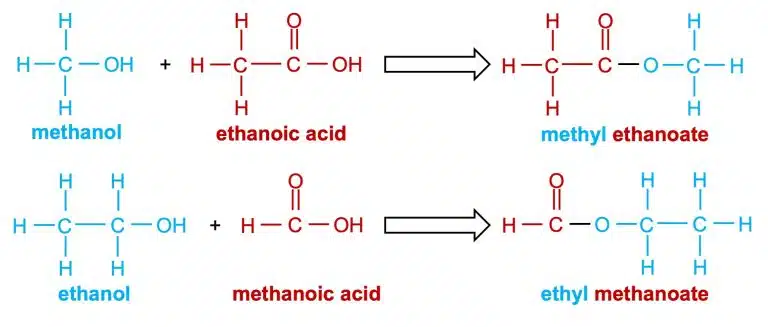
Esters are named after their parent alcohol and carboxylic acid. For example, the ester formed between methanol and ethanoic acid is called methyl ethanoate, while the ester formed between ethanol and methanoic acid is called ethyl methanoate.
Esters have many uses in the chemical industry: they are used in artificial flavourings and fragrances, as solvents for paints, glues and nail polish remover, and as an ingredient in soaps and cosmetics.

Many essential molecules that support our biological functions are esters as well. The majority of fats or lipids in our bodies and in the food we consume are triglycerides: esters made from glycerol and three fatty acids.
We hope that this post not only helped you to review the key homologous series in the ‘O’-Level syllabus, but also brought you a new perspective about the sheer diversity and versatility of the organic molecules and reactions occurring around us every day.
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us