- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

Before you read on, you might want to download this entire revision notes in PDF format to print it out for your child, or yourself to read it later.
This will be delivered to your email inbox.
In the study of Chemistry, understanding energy is just as vital as understanding the atoms themselves.
Often referred to as the “Central Science,” Chemistry links the physical laws of energy to the biological processes of life.
When we discuss energy in the context of the Singapore GCE O Level syllabus, we are primarily focused on chemical energetics which is referring to the study of how energy is absorbed or released during chemical reactions.
To understand this topic, you must first understand that every chemical reaction involves a change in energy because chemical bonds act as “energy stores”.
To break a bond, energy must be absorbed, and when new bonds are formed, energy is released.
Mastering this topic is not about rote memorization; it is about recognizing the patterns of how matter and energy interact to shape the world around us.
To maximize your performance in the Singapore GCE O Level Chemistry examination, this guide should be treated as a structured revision checklist rather than a simple list to be memorized.
Success in Chemical Energetics depends on your ability to recognize patterns in how energy is absorbed or released during chemical reactions.
You should start by understanding the Fundamentals which refers to things like the distinction between bond breaking (endothermic), and bond forming (exothermic).
When preparing for the practical component, which can be the difference between an A1 and an A2, ensure you record temperature data to a precision of one decimal place using a thermometer. ALWAYS, use the POD framework (Procedure, Observation, and Data Processing) to practice writing experimental designs for enthalpy changes, ensuring your instructions are imperative and specific with quantities.
Additionally, consult the Common Mistakes section to refine your evaluation skills.
For example, always suggest using a polystyrene cup or a lid to minimize heat loss to the surroundings rather than citing vague “human error”.
Finally, verify that your calculated data matches the precision of your measured data to ensure you meet the technical rigor demanded by examiners.
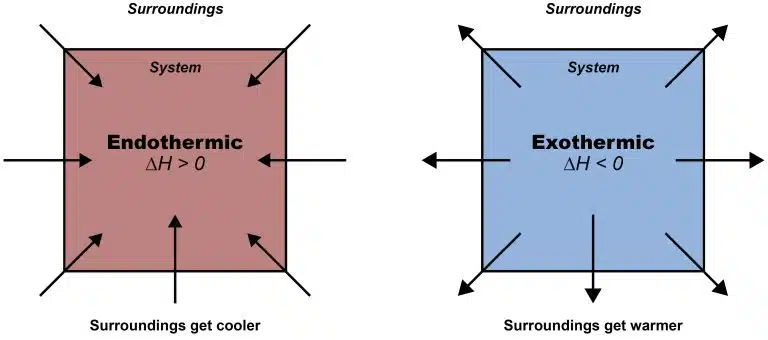
The core concept of energy in GCE O Levels is the concept of exothermic and endothermic reaction. This is basically your foundation towards everything you will encounter in your paper.
The terms exothermic and endothermic reactions, refer to the direction of heat flow between the reaction and its surroundings. But how do we understand these terms? Like all words, usually the prefix does tell us something about the meaning of the word. We can look at the table below to “guess” the meaning of endothermic and exothermic.
TERM | Similar Terms | Meaning |
Endothermic | EN-velope: To take in something inside the envelope | The meaning of endothermic is to absorb/take in energy from the surroundings. |
Exothermic | EX-pel: To release something | The meaning of exothermic is to release energy to the surroundings. |
An exothermic reaction is defined as a chemical process that releases heat energy to its environment. This typically results in a measurable temperature rise in the reacting solution or the immediate surroundings. Because the products in an exothermic reaction possess less energy than the reactants, the enthalpy change is always negative ( ).
Common examples include neutralization between acids and bases, the combustion of fuels, and the oxidation of metals which combines a metal with oxygen to form a solid metal oxide.
In contrast, an endothermic reaction is a process where the system absorbs heat energy from its surroundings. This absorption causes the temperature of the environment to drop. In these reactions, the products have more energy than the reactants, resulting in a positive enthalpy change ( ).
A classic example is thermal decomposition, where a single compound breaks down into simpler substances only when heat is continuously applied. For instance, heating calcium carbonate to produce calcium oxide and carbon dioxide is a staple endothermic reaction in the syllabus.
To understand why a reaction is exothermic or endothermic, one must look at the internal “energy balance” of chemical bonds.
All chemical reactions involve the absorption of energy to break existing bonds in the reactants and the release of energy when new bonds are formed in the products. The overall energy change of the reaction is determined by the difference between these two quantities.
If the energy released during bond formation is greater than the energy absorbed during bond breaking, the reaction is exothermic.
Conversely, if more energy is required to break the bonds than is released when new ones form, the reaction is endothermic.
In the practical component of the O Level, you will often be asked to determine the maximum temperature change of a reaction.
Recording this data requires scientific precision; for example, temperatures must be recorded using a thermometer to one decimal place, typically ending in .0 or .5.
A frequent challenge in energy experiments is the loss of heat to the surroundings, which can lead to inaccurate results.
When evaluating these experiments, students should avoid vague terms like “human error”. Instead, specific technical improvements should be suggested, such as insulating the reaction vessel with polystyrene or using a lid to minimize environmental heat transfer.
When describing these observations in Qualitative Analysis, always use standard phrasing: describe the formation of precipitates or the specific results of gas tests, such as hydrogen extinguishing a lighted splint with a “pop” sound, rather than using casual language.
When tasked with planning an energy-related experiment, the “POD” framework: Procedure, Observation, and Data Processing, is essential for securing full marks.
The procedure should be written in imperative, numbered steps, being highly specific with quantities, such as measuring 20ml of acid using a measuring cylinder. Observations must explicitly state what tool is being used for measurement, such as a stopwatch for timing or a thermometer for temperature.
Finally, data processing explains how the raw measurements will be used to answer the experimental aim, often involving the plotting of a graph or calculating a temperature change. Students often forget to account for these in their answers.
Your written answers also require avoiding “forbidden” words that lack scientific rigor. For example, “amount” is considered too vague and should be replaced with “mass” for solids or “volume” for liquids and gases.
Similarly, a liquid should be described as “colorless” rather than “clear,” and substances are said to “dissolve” or “react completely” rather than “disappear”.
By adhering to these conventions and mastering the energy balance of chemical reactions, you will be well-prepared to achieve a distinction in O Level Chemistry.
Mastering chemical energetics is a fundamental milestone in the Singapore GCE O Level Chemistry syllabus, serving as a gateway to understanding how the world functions at a molecular level.
As we have explored throughout this guide, the study of energy changes, from the exothermic release of heat in combustion to the endothermic absorption of energy in thermal decomposition is governed by the immutable balance of bond breaking and bond forming.
Achieving an A1 in this topic requires more than just understanding these theoretical concepts; it demands a high level of technical rigor and precision in both your written and practical work.
Furthermore, your performance in the practical component (Paper 3) relies on your ability to record raw data, such as thermometer readings, to a consistent one decimal place and to critically evaluate experimental setups by suggesting specific improvements like polystyrene insulation instead of citing vague human error.
By utilizing the POD framework for planning and adhering to the strict formatting conventions for tables and graphs, you transform from a student who simply knows chemistry into one who can professionally communicate scientific findings.
Treat every chemical equation as a recipe for energy transfer and walk into your examination hall with the confidence that comes from mastering the precise language of chemical energetics.
Once again, best of luck for your revision for O Level Chemistry!
Find out more by joining us at Science of Studying!
Prepared by: Lys
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us