- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

The reactivity series is a vital concept in O-Level Chemistry that ranks metals based on their reactivity with other substances, such as acids and water. Understanding the reactivity series is crucial for various applications, including metal extraction, corrosion prevention, and displacement reactions.
In this article, we will delve into the characteristics of the reactivity series, its significance, and how it influences chemical behavior in practical situations.
But before you go on reading… You might want to download a pdf copy of this article as it is quite long!
Click the ‘Download’ button, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)

The metal reactivity series is a list of metals arranged in order of their reactivity, starting with the most reactive and ending with the least reactive. The position of a metal in this series determines how it reacts with water, acids, and other substances.
Mnemonic | Reactivity | ||
Potassium (K) | Please | React with water | ^ ^ |
Sodium (Na) | Stop | ||
Calcium (Ca) | Calling | ||
Magnesium (Mg) | Me | React with acid | |
Aluminium (Al) | A | ||
(Carbon) | (Cool) | ||
Zinc (Zn) | Zebra | ||
Iron (Fe) | I | ||
Lead (Pb) | Like | ||
(Hydrogen) | (Happy) | ||
Copper (Cu) | Cute | Unreactive | |
Silver (Ag) | Ant | ||
For O-Level Pure Chemistry, understanding the series helps you predict which metals can displace others in reactions and how they will interact with different chemicals.
A more reactive metal can displace a less reactive metal from its compound. For example, if you place a strip of zinc in a solution of copper(II) sulfate (CuSO4), zinc will displace the copper:
Zn + CuSO4 → ZnSO4 + Cu
This series not only helps predict the outcomes of chemical reactions but also provides insights into the properties and behaviors of different metals.
Metals | Reaction with Cold Water | Reaction with Steam | Reaction with Acid | Thermal Decomposition of Carbonates |
Metal + cold water → metal hydroxide + hydrogen | Metal + steam → metal oxide + hydrogen | Metal + acid → salt + hydrogen | Metal carbonates → metal oxide + carbon dioxides OR metal + oxygen + carbon dioxides | |
Potassium | React violently to form metal hydroxide and hydrogen gas | React explosively to form metal oxide and hydrogen gas | React explosively to form salt and hydrogen gas | Does not decompose (stable to heat) |
Sodium | ||||
Calcium | React rapidly to form salt and hydrogen gas | Decompose to form metal oxide and carbon dioxide gas | ||
Magnesium | React slowly (since magnesium hydroxide acts as protective barrier) | React violently to form magnesium oxide and hydrogen gas | ||
Aluminium | No reaction (since aluminium hydroxide acts as protective barrier) | No reaction (since aluminium oxide acts as protective barrier) | ||
(Carbon) | ||||
Zinc | No reaction | React readily to form zinc oxide and hydrogen gas | React quickly to form zinc salt and hydrogen gas | Decompose to form metal oxide and carbon dioxide gas |
Iron | React slowly to form iron (II/ III) oxide and hydrogen gas | Reacts slowly to form iron (II/III) salt and hydrogen gas | ||
Lead | No reaction | No reaction (since PbCl2 is insoluble even though above H) | ||
(Hydrogen) | ||||
Copper | No reaction | No reaction | No reaction | Decompose to form metal oxide and carbon dioxide gas |
Silver | Decomponse to form metal, oxygen and carbon dioxide gas | |||
Gold | ||||
Platinum |
The extraction of metals from their ores varies depending on their position in the metal reactivity series. Metals higher in the series are more reactive and require more energy-intensive methods of extraction, while those lower in the series can be extracted more easily.
Metals | Extraction Method |
Potassium | Electrolysis |
Sodium | |
Calcium | |
Magnesium | |
Aluminium | |
(Carbon) | |
Zinc | Reduction of metal oxides with carbon |
Iron | |
Lead | |
(Hydrogen) | |
Copper | Reduction of metal oxides with hydrogen |
Silver | |
Gold | Found in native state |
Platinum |
Let’s look at a common example of metal extraction below.
Iron is one of the most widely used metals in the world. The extraction of iron using carbon is a process that has been practiced for centuries, forming the basis of steel production, which is vital for industries ranging from construction to manufacturing.
The extraction of iron using carbon occurs in a large furnace known as a blast furnace, which is designed to withstand extremely high temperatures. The key materials used in this process are:
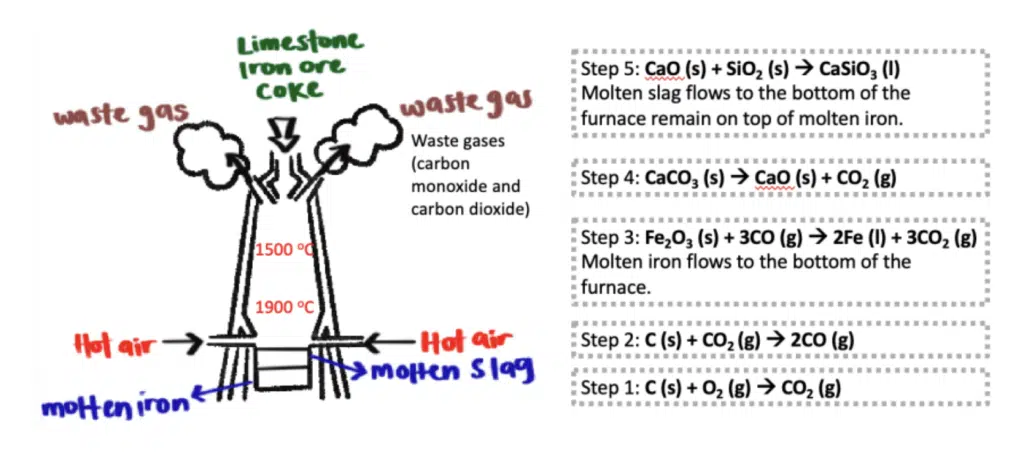
As we delve into the topic of iron, it’s essential to also explore the phenomenon of rusting.
Rusting is a common and destructive chemical process that occurs when iron is exposed to moisture (H2O) and air (O2) over time. This process leads to the formation of a reddish-brown substance known as rust, which can weaken and damage iron structures and objects.
Iron + Oxygen + Water → Hydrated iron (III) oxide
rust
4Fe(s) + 3O2(g) + 2xH2O(l) → 2Fe2O3.xH2O(s)
Given the damaging effects of rust, it is important to employ methods to prevent or slow down the rusting process. Here are some common strategies:
Methods | Description | Uses |
Barrier (e.g., painting, coating with grease/oil) | Applying a layer of paint or oil provides a physical barrier that prevents moisture and oxygen from coming into direct contact with the iron | Cars, bridges, machines |
Alloying (e.g., stainless steel) | Alloying iron with other metals enhances its resistance to rust. Chromium in stainless steel forms a thin, stable oxide layer on the surface that prevents further corrosion | Cutleries |
Galvanising | Coating iron or steel with a layer of zinc, which acts as a sacrificial metal, meaning it corrodes in place of the iron | Buildings, signboards, sculptures |
Sacrificial protection | Iron is connected to a more reactive metal, such as magnesium or zinc, which acts as a sacrificial anode. The more reactive metal corrodes instead of iron, thereby protecting it from rust. | Pipelines, water heaters, ship hulls |
In conclusion, the reactivity series provides valuable insights into the behavior of metals in various chemical reactions and in practical applications, such as metal extraction and the prevention of corrosion. Mastery of the reactivity series will not only enhance your problem-solving skills, but also prepare you for advanced topics in chemistry. Keep exploring the fascinating world of chemistry, and let your curiosity guide you in discovering how these principles manifest in everyday life.
Find this helpful? Find out more by joining us at Science of Studying!
Prepared by: Tan Wee Leng
This article is written based on the 6092 Chemistry GCE Ordinary Level Syllabus. https://www.seab.gov.sg/docs/default-source/national-examinations/syllabus/olevel/2024syllabus/6092_y24_sy.pdf
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
At Science of Studying, we provide live Zoom-based online tuition for Combined Chemistry, Combined Biology, Pure Science subjects, and JC-level Chemistry and Biology. We’ve had the privilege of teaching over 800 students across 12 years.
Our SOS system™️ gives students a clear, effective way to learn – helping even last-minute learners improve quickly, without relying on memorising entire textbooks or grinding through excessive practice papers. All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry and Biology subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us