- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

Before you read on, you might want to download this entire revision notes in PDF format to print it out for your child, or yourself to read it later.
This will be delivered to your email inbox.
Chemistry is frequently referred to as the “Central Science” because it links biology and physics. However, its distinctive language, chemical equations, poses a problem for a lot of students.
Equations are the universe’s recipes, not just collections of letters and numbers that must be committed to memory. They specify what products are produced, what reactants are needed, and the exact circumstances needed to make it happen. Mastering O level chemistry requires you to be fluent in this language. You need to be able to translate a word problem into a chemical formula, balance the atoms, and calculate the masses involved.
Do not try to memorize every single equation in one night. Instead, focus on the patterns.
If you learn the general pattern for “Acid + Carbonate,” you don’t need to memorize fifty different specific reactions; you just apply the pattern to the specific chemicals in the question.
Use this guide as your checklist. If you can look at each section and confidently write out the equations from memory, you are ready for your O Levels.
In the GCE O Level syllabus, there are a few common acids used, these acids are hydrochloric acid, nitric acid and sulfuric acid. The acid used determines the second word of the “Salt” name.
Example:
Acid used | Second Word of the “Salt” name | Examples of salts |
Hydrochloric Acid (𝐻𝐶𝑙) | Chloride | Magnesium Chloride Sodium Chloride Aluminum Chloride |
Sulfuric Acid (𝐻₂𝑆𝑂₄) | Sulfate | Potassium Sulfate Magnesium Sulfate Aluminum Sulfate |
Nitric Acid (𝐻𝑁𝑂₃) | Nitrate | Potassium Nitrate Sodium Nitrate Aluminum Nitrate |
Example:
Acid used | Reactant used | Salt Produced |
Hydrochloric Acid (𝐻𝐶𝑙) | Magnesium | Magnesium Chloride |
Magnesium Hydroxide | ||
Magnesium Carbonate | ||
Sulfuric Acid (𝐻₂𝑆𝑂₄) | Sodium | Sodium Sulfate |
Sodium Oxide (Metal Oxides are Basic) | ||
Sodium Hydroxide | ||
Sodium Carbonate |
Neutralization is a chemical reaction in which an acid and a base (or alkali) react quantitatively to form a salt and water.
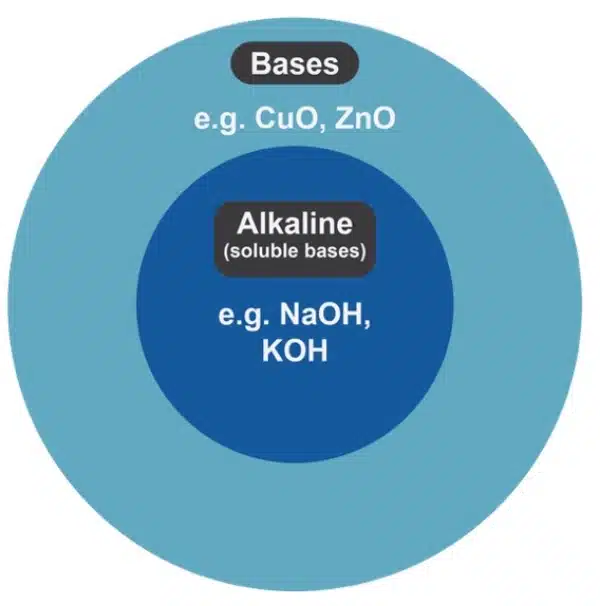
Bases and alkalis are similar, but not the same. Certain textbooks use the two words interchangeably but here is why they are not the same.
A base is any substance that can neutralize an acid (react with it to form salt and water). An alkali, however, is a special type of base.
The key difference is water solubility: an alkali is a base that dissolves in water.
In other words,
All alkalis are bases, but not all bases are alkalis.
For the GCE O Level Syllabus, bases commonly include both metal oxides and metal hydroxides whereas alkali only include metal hydroxides. This is extremely crucial when writing chemical equations.
In an aqueous solution, the essential reaction occurs between the hydrogen ions from the acid and the hydroxide ions, OH– from the alkali to produce neutral water molecules.
The ionic equation for this process is:
𝐻⁺ + 𝑂𝐻– → 𝐻₂𝑂
The general equation for this process is:
𝐴𝑐𝑖𝑑 + 𝐵𝑎𝑠𝑒 → 𝑆𝑎𝑙𝑡 + 𝑊𝑎𝑡𝑒𝑟
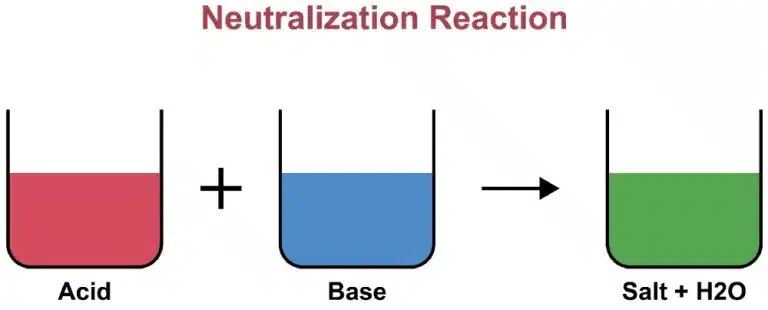
This reaction typically moves the pH of the solution toward 7 and is often exothermic (releases heat).
A common example is the reaction between hydrochloric acid and sodium hydroxide.
𝐻𝐶𝑙 (𝑎𝑞) + 𝑁𝑎𝑂𝐻 (𝑎𝑞) → 𝐻₂𝑂 (𝑙) + 𝑁𝑎𝐶𝑙 (𝑎𝑞)
In everyday life, neutralization is vital for applications such as treating acidic soil with lime (calcium hydroxide) or relieving acid indigestion with antacid tablets.
An acid-metal reaction is a chemical process where an acid reacts with a metal to produce a salt and hydrogen gas. In experimental chemistry, this is often the reaction responsible for the “fizzing” or effervescence seen when metals are placed in acid.
The general equation for this process is:
𝑚𝑒𝑡𝑎𝑙 + 𝑎𝑐𝑖𝑑 → 𝑠𝑎𝑙𝑡 + ℎ𝑦𝑑𝑟𝑜𝑔𝑒𝑛 𝑔𝑎𝑠
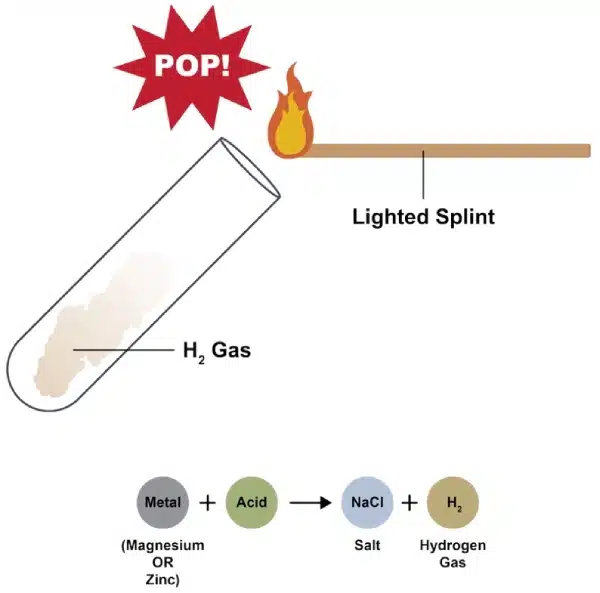
Key Observation: The production of Hydrogen gas is the defining feature. This can be tested using a lighted splint; if hydrogen is present, it will extinguish with a characteristic “pop” sound.
However, a common misconception is that acids react with all metals. This is not true. Acids only react with metals that are more reactive than hydrogen in the reactivity series. In the syllabus, there are 3 common metals which are unreactive with acid, these metals are Copper, Silver and Gold.
To illustrate this, acids will react vigorously with Magnesium or Zinc, but they will not react at all with Copper, Silver, or Gold.
This reaction occurs when an acid reacts with a metal carbonate.
Unlike the acid-metal reaction, this process produces three products: a salt, water, and carbon dioxide gas.
The general equation for this process is:
𝐴𝑐𝑖𝑑 + 𝐶𝑎𝑟𝑏𝑜𝑛𝑎𝑡𝑒 → 𝑆𝑎𝑙𝑡 + 𝑊𝑎𝑡𝑒𝑟 + 𝐺𝑎𝑠
An example of this reaction between hydrochloric acid and sodium carbonate is also shown below:
𝐻𝑦𝑑𝑟𝑜𝑐ℎ𝑙𝑜𝑟𝑖𝑐 𝐴𝑐𝑖𝑑 + 𝑆𝑜𝑑𝑖𝑢𝑚 𝐶𝑎𝑟𝑏𝑜𝑛𝑎𝑡𝑒 → 𝑆𝑜𝑑𝑖𝑢𝑚 𝐶ℎ𝑙𝑜𝑟𝑖𝑑𝑒 + 𝐶𝑎𝑟𝑏𝑜𝑛 𝐷𝑖𝑜𝑥𝑖𝑑𝑒 + 𝑊𝑎𝑡𝑒𝑟
2𝐻𝐶𝑙 (𝑎𝑞) + 𝑁𝑎₂𝐶𝑂₃ (𝑠) → 2𝑁𝑎𝐶𝑙 (𝑎𝑞) + 𝐶𝑂₂ + 𝐻₂𝑂
This reaction is extremely useful in the lab for identifying unknown powders. If you add acid to a substance and it produces effervescence, we can test for the presence of carbon dioxide by bubbling the gas into limewater, 𝐶𝑎(𝑂𝐻)₂.
. If a white precipitate is formed, carbon dioxide is present.
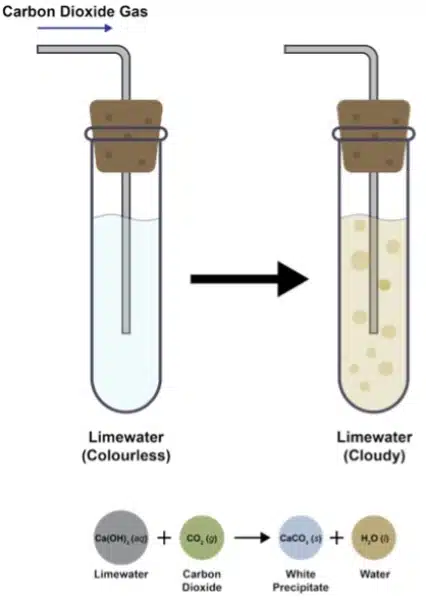
Key Observation:
The effervescence observed is Carbon Dioxide (𝐶𝑂₂) gas escaping. To confirm the gas’s identity, the gas is bubbled through limewater
𝐶𝑎(𝑂𝐻)₂.
If 𝐶𝑂₂ is present, the limewater forms a white precipitate 𝐶𝑎𝐶𝑂₃.
The equation for the limewater test is frequently tested and shown below:
𝐶𝑎 (𝑂𝐻)₂ (𝑎𝑞) + 𝐶𝑂₂ (𝑔) → 𝐶𝑎𝐶𝑂₃ (𝑠) + 𝐻₂𝑂 (𝑙)
Combustion is a chemical reaction in which a substance (Either a hydrocarbon or a fuel) reacts rapidly with oxygen and gives off heat. This reaction is exothermic (release heat energy).
There are two different types of combustion:
1. Complete combustion: Which occurs when there is excess oxygen. This produces Carbon Dioxide and Water.
2. Incomplete combustion: Which occurs when there is insufficient oxygen. This produces Carbon Monoxide and Water.
When a metal is combusted under excess oxygen, it follows the equation below:
𝐻𝑦𝑑𝑟𝑜𝑐𝑎𝑟𝑏𝑜𝑛 + 𝑂𝑥𝑦𝑔𝑒𝑛 ![]() 𝐶𝑎𝑟𝑏𝑜𝑛 𝐷𝑖𝑜𝑥𝑖𝑑𝑒 + 𝑊𝑎𝑡𝑒𝑟
𝐶𝑎𝑟𝑏𝑜𝑛 𝐷𝑖𝑜𝑥𝑖𝑑𝑒 + 𝑊𝑎𝑡𝑒𝑟
Elsewise, when under limited oxygen, it follows the equation below:
𝐻𝑦𝑑𝑟𝑜𝑐𝑎𝑟𝑏𝑜𝑛 + 𝐿𝑖𝑚𝑖𝑡𝑒𝑑 𝑂𝑥𝑦𝑔𝑒𝑛 ![]() 𝐶𝑎𝑟𝑏𝑜𝑛 𝑀𝑜𝑛𝑜𝑥𝑖𝑑𝑒 + 𝑊𝑎𝑡𝑒𝑟
𝐶𝑎𝑟𝑏𝑜𝑛 𝑀𝑜𝑛𝑜𝑥𝑖𝑑𝑒 + 𝑊𝑎𝑡𝑒𝑟
Thermal decomposition is a chemical reaction where a single compound breaks down into two or more simpler substances when heated.
Unlike combustion, which releases heat, thermal decomposition is endothermic, it absorbs heat energy from the surroundings to break the chemical bonds.
This means the reaction will stop if you stop heating it.
The general equation for the thermal decomposition of a metal carbonate is:
𝑀𝑒𝑡𝑎𝑙 𝐶𝑎𝑟𝑏𝑜𝑛𝑎𝑡𝑒 ![]() 𝑀𝑒𝑡𝑎𝑙 𝑂𝑥𝑖𝑑𝑒 + 𝐶𝑎𝑟𝑏𝑜𝑛 𝐷𝑖𝑜𝑥𝑖𝑑𝑒
𝑀𝑒𝑡𝑎𝑙 𝑂𝑥𝑖𝑑𝑒 + 𝐶𝑎𝑟𝑏𝑜𝑛 𝐷𝑖𝑜𝑥𝑖𝑑𝑒
A classic example in the syllabus is the heating of calcium carbonate (limestone) to make quicklime. This can be represented by the word and chemical equations below.
𝐶𝑎𝑙𝑐𝑖𝑢𝑚 𝐶𝑎𝑟𝑏𝑜𝑛𝑎𝑡𝑒 ![]() 𝐶𝑎𝑙𝑐𝑖𝑢𝑚 𝑂𝑥𝑖𝑑𝑒 + 𝐶𝑎𝑟𝑏𝑜𝑛 𝐷𝑖𝑜𝑥𝑖𝑑𝑒
𝐶𝑎𝑙𝑐𝑖𝑢𝑚 𝑂𝑥𝑖𝑑𝑒 + 𝐶𝑎𝑟𝑏𝑜𝑛 𝐷𝑖𝑜𝑥𝑖𝑑𝑒
𝐶𝑎𝐶𝑂₃ (𝑠) ![]() 𝐶𝑎𝑂 (𝑠)+𝐶𝑂₂ (𝑔)
𝐶𝑎𝑂 (𝑠)+𝐶𝑂₂ (𝑔)
Since Carbon Dioxide is released in this reaction, we can also test for the gas using limewater, 𝐶𝑎(𝑂𝐻)₂.
. Bubble the gas into limewater, and if carbon dioxide is produced, a white precipitate is observed.
However, the “burning” of a metal is slightly different. This reaction is still considered combustion as it releases heat and light, but it is chemically distinct from burning fuels like hydrocarbon and wood (Reaction 4). Unlike the combustion of fuels, the combustion of metal doesn’t produce any carbon dioxide or water as this reaction is a direct reaction with oxygen. The combustion of metal can thus be considered as oxidation.
The reaction is a synthetical reaction where metal combines with oxygen from the air to form a solid metal oxide.
It is represented by a general equation below:
𝑀𝑒𝑡𝑎𝑙+𝑂𝑥𝑦𝑔𝑒𝑛 → 𝑀𝑒𝑡𝑎𝑙 𝑂𝑥𝑖𝑑𝑒
An example of the word and chemical equation for the combustion of Magnesium Metal is also shown below:
𝑀𝑎𝑔𝑛𝑒𝑠𝑖𝑢𝑚 + 𝑂𝑥𝑦𝑔𝑒𝑛 → 𝑀𝑎𝑔𝑛𝑒𝑠𝑖𝑢𝑚 𝑂𝑥𝑖𝑑𝑒
2𝑀𝑔 (𝑠) + 𝑂₂(𝑔) → 2𝑀𝑔𝑂 (𝑠)
Key Observation: The most striking feature of this reaction is often a change in mass. Because oxygen atoms from the air are physically attaching themselves to the metal to form the oxide, the mass of the solid increases after combustion.
Plants grow best in soil that is neither too acidic nor too alkaline (usually pH 6–7). However, due to acid rain and overuse of fertilizers, there is a potentiality of soil becoming too acidic. Since this is not ideal for the growth of the plants, farmers must then neutralize it, with a process known as liming.
This involves adding basic compounds, specifically Carbonates and Metal Oxides or Alkalis, to react with the excess hydrogen ions caused by the acidic soil.
RECALL: Acid-Base Reactions and Acid-Carbonate Reactions
The purpose of liming is to get rid of excess acid, and thus, we must add substances which are able to react with acids, except for metals.
The most common material used is crushed Limestone, which is Calcium Carbonate, . Since it is a carbonate, it reacts with the acid via an acid-carbonate reaction to produce a salt, water, and carbon dioxide gas.
The equation between Calcium carbonate and Hydrochloric acid is shown below.
𝐶𝑎𝐶𝑂₃(𝑠) + 2𝐻𝐶𝑙 (𝑎𝑞) → 𝐶𝑎𝐶𝑙₂(𝑎𝑞) + 𝐻₂𝑂 (𝑙) + 𝐶𝑂₂ (𝑔)
If a faster change is needed, farmers may use Slaked Lime (𝐶𝑎(𝑂𝐻)₂).
This is an alkali because it is slightly soluble in water, releasing hydroxide ions, that react immediately with acidic soil.
Article written by: Teacher Lys
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us