- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

Before you read on, you might want to download this entire revision notes in PDF format to print it out for your child, or yourself to read it later.
This will be delivered to your email inbox.
If you are on this page, there is a very good chance that the device that you are reading this article on has components made of polymers.
In fact, it is also likely that the clothes you are wearing, the water cup or bottle in your bag or on your desk, and the case protecting your phone are made in some part out of polymers. The majority of synthetic materials in our lives are produced by a process called polymerisation – but what exactly is this process?
Let’s explore the world of polymers together in this article.
Polymers are macromolecules – very large molecules that are made by linking many smaller molecules together. These smaller molecules are known as monomers, and by changing the number and type of monomers used, polymers with different molecular weights and properties can be produced.

Many identical monomer molecules will react together to form a polymer, and they form repeating units within the polymer chain. A polymer can be made of one or more types of monomers.
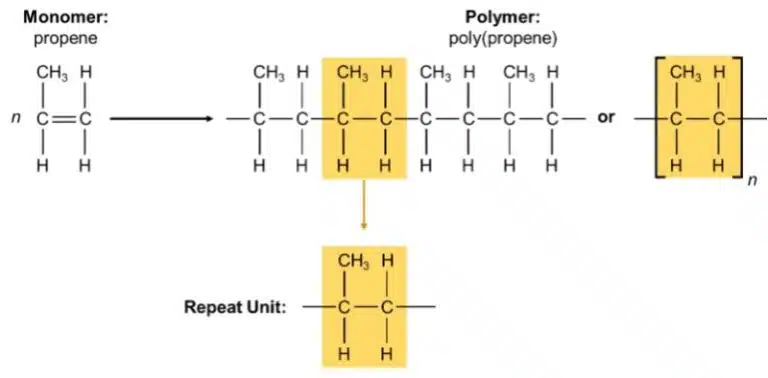
There are two types of polymerisation reactions to know for ‘O’-Levels: addition polymerisation and condensation polymerisation.
During addition polymerisation, monomers join together to form a polymer with no other side products. Addition polymers are made from monomers with C=C double bonds, i.e. alkenes; during polymerisation, the double bond ‘opens up’ and connects with other monomer molecules to form the polymer chain.
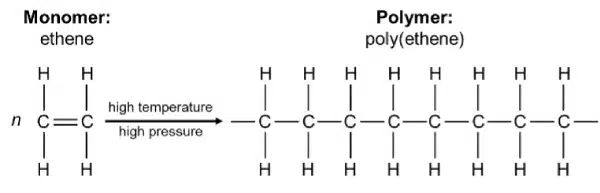
Since the C=C double bonds are broken during polymerisation, the resulting polymer is saturated and will not decolourise aqueous bromine.
Many plastic objects we use in our daily lives are in fact made from addition polymers; examples include poly(ethene) or polyethylene (PE), poly(propene) or polypropylene (PP), and poly(vinyl chloride) (PVC).
Poly(ethene) is waterproof, lightweight, resistant to corrosion and inexpensive, hence it is commonly used to make shopping bags, cling film, food containers and bottles. However, as C-H and C-C bonds are unreactive to most reagents, it is a non-biodegradable material and contributes significantly to land and water pollution.

In condensation polymerisation, monomers react together to form polymer chains, as well as another small molecule as a side product. The main condensation polymers we study in ‘O’-Level Chemistry are polyesters and polyamides.
Terylene, a polyester
If you pay close attention to the laundry tags on your clothes, chances are you’ll find several garments that are completely or partially made from polyester fibres. A polyester is a polymer that is held together by ester linkages, which we have learnt can be formed between alcohol groups and carboxylic acid groups.

Let’s look at the case study of Terylene, which is the trade name for poly(ethylene terephthalate) or PET. Terylene is made from two monomers, a diol, which has two alcohol groups, and a dicarboxylic acid, which has two carboxylic acid groups.
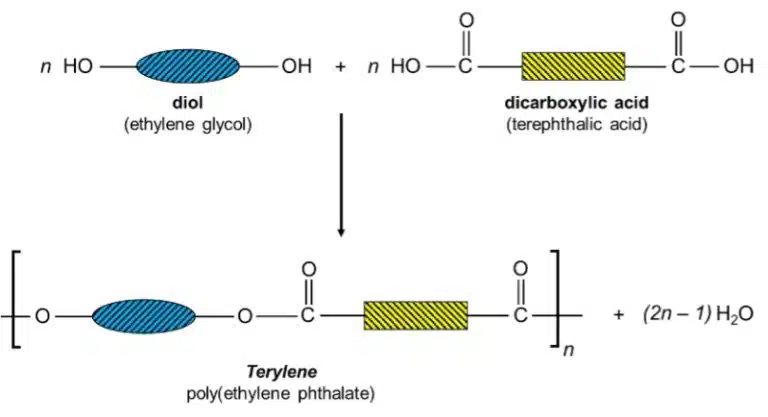
The diol and dicarboxylic acid molecules undergo esterification to form a long polymer chain held together by ester linkages. A water molecule is given out for each ester linkage formed.
Terylene fabric resists stretching and wrinkling, and is used to make clothing, curtains, among other fabric goods. It is also flame-resistant as it is difficult to ignite, so it can also be used in protective suits for firefighters and rescue operators.

Nylon was one of the very first synthetic fibres marketed to the public; nylon stockings were widely publicised by manufacturer DuPont at the 1938 New York World’s Fair, before hitting store shelves in May of 1940. The stockings were an instant hit; they sold out at noon at most retail locations on the first day they became available, and within two years DuPont had captured 30% of the entire American hosiery market. Stockings were an essential part of everyday dress for women at the time, yet silk was expensive, dainty and increasingly difficult to import due to wartime tensions; nylon offered a more durable, elastic and affordable alternative.
When DuPont eventually turned over its entire nylon production to the military for parachutes and ropes, stockings became a luxury that commanded exorbitant prices on the black market; women took great care to maintain their remaining stockings, and even resorted to painting over their legs with makeup, shoe polish, tea or even gravy to avoid appearing bare-legged in public – a major taboo at the time. “Nylons” finally returned to the market in 1945, causing ‘riots’ wherever they were stocked; one store reported mile-long queues of 40,000 customers competing for 13,000 pairs.

Nylon is an example of a polyamide – a condensation polymer held that is held together by amide linkages. An amide is an organic compound that contains the ─C=ONH─ amide group (as shown below). Amides can be produced by reacting carboxylic acids with amines, which are molecules that contain the ─NH2 group.
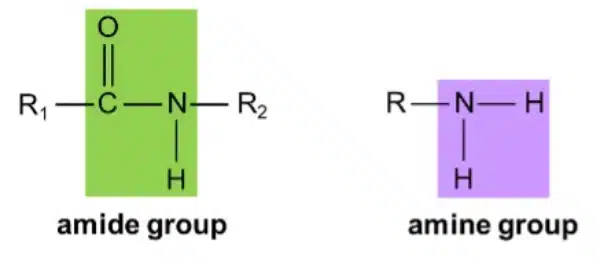
Like Terylene, nylon is made of two different monomers: a dicarboxylic acid, and a diamine, which is a molecule with two amine groups. The carboxylic acid groups and amine groups undergo condensation to form amide linkages, resulting in a long polyamide chain.
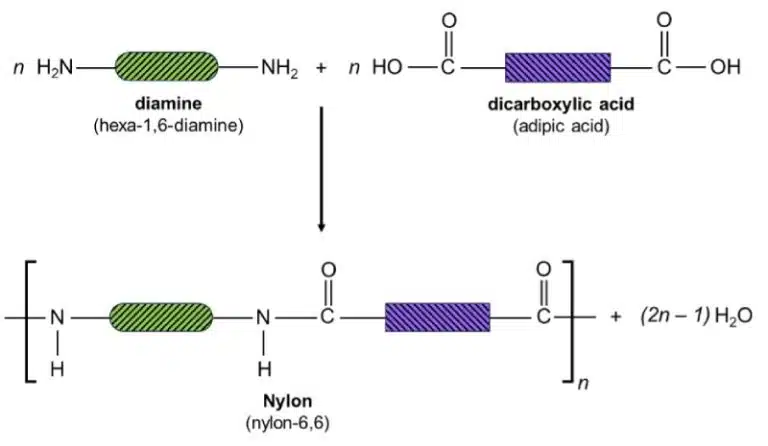
Nylon is strong and durable, and is still widely used to make many products today. Toothbrush bristles, fishing lines and raincoats are just a few examples of everyday objects that are made from nylon.

One of the most useful properties of polymers is that they are unreactive and durable, which means they can be used to make food-safe containers, utensils and wrappings that last a long time.
However, the chemical resistance of plastics makes them more difficult to dispose of – many plastics, especially addition polymers such as poly(ethene) and poly(propene) are non-biodegradable, which means that micro-organisms are unable to break them down. As a result, plastic waste takes up a great amount of space in landfills, and can remain in the earth for many hundreds of years.
While it is possible to incinerate plastics and use the heat generated to produce electricity or provide heating to homes, this waste disposal method has its own disadvantages: it releases carbon dioxide which is a greenhouse gas, and also toxic waste gases such as carbon monoxide (due to incomplete combustion), sulfur dioxide, nitrogen oxides and hydrogen chloride (if the polymer contains sulfur, nitrogen or chloride atoms).

It is possible to recycle most forms of plastic waste; however, as plastic objects may be made of different types of polymers with vastly different treatment methods, they must be separated beforehand.
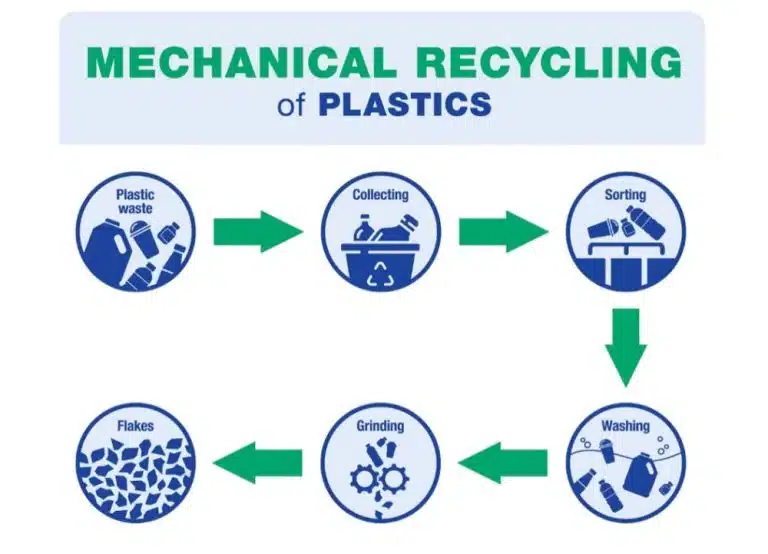
In general, there are two methods to recycle plastics: physical and chemical. In physical recycling (also known as mechanical recycling), waste plastic is ground up into smaller pieces and melted into pellets, which can be repurposed to make new objects. For example, a plastic bottle made of PET (a polyester) can be shredded and melted into small pellets, which are extruded into threads that can be woven into fabric for shoes.
In chemical recycling, on the other hand, the covalent bonds within the polymer molecules are broken up by chemical reactions. This can be done to recover the original monomers, such as in depolymerisation, or to produce completely different molecules entirely, such as through cracking of poly(ethene) to obtain chemical feedstock and fuel.
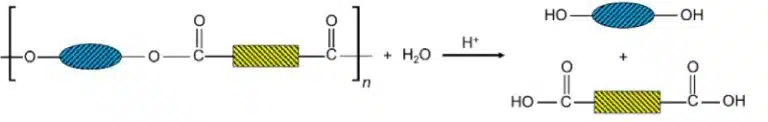
Polyesters and polyamides can be depolymerised and broken down back into their monomers, as both ester and amide linkages can be hydrolysed by strong acids or alkalis.
Concerns about plastic waste have given rise to a growing market for biodegradable plastics – polymers that are designed to be broken down by micro-organisms within a few decades. Since many plastic monomers are derived from crude oil, there is also great interest in bioplastics which are produced from renewable sources, such as polylactic acid (PLA) from milk or fermented plant starch.
Polymers, especially in the form of plastics, are an inescapable part of our modern lives due to the versatility of their properties and their durability. Hence, as chemistry students, it is important for us to understand how they are produced from monomers and think about how they can be disposed of responsibly.
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us