- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

The periodic table is one of the most powerful tools in chemistry, organising the elements in a way that reveals patterns in their properties and behaviors. For A-Level H2 Chemistry students, a deep understanding of the periodic table is essential, as it underpins much of the subject’s advanced concepts, from atomic structure to chemical bonding and reactivity trends.
In this article, let’s delve deeper into the trends and variations of atomic, physical, and chemical properties.
Click here for O Levels Period Table Guide.
But before you go on reading… You might want to download a pdf copy of this article as it is quite long!
Click the ‘Download’ button, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)

The modern periodic table is arranged in order of increasing atomic number (the number of protons in an atom’s nucleus). The elements are organized into rows called periods and columns called groups.
Across a period, elements have
Down a group, elements have
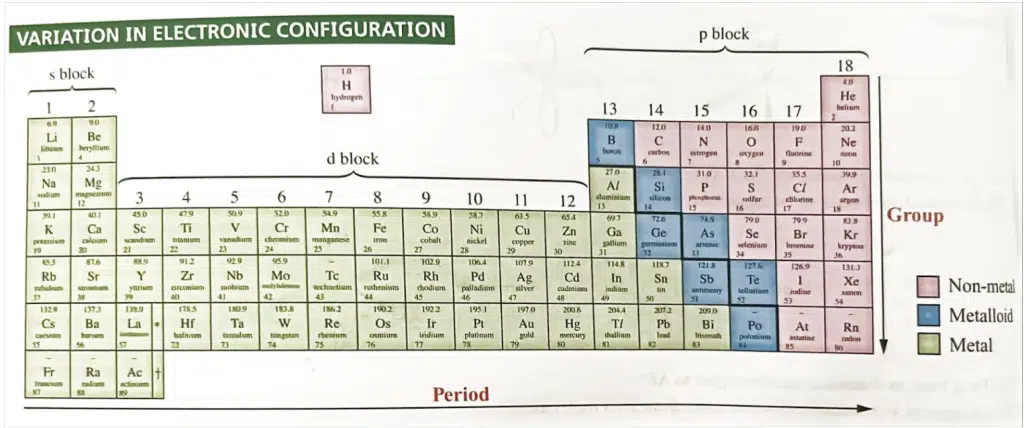
Several key periodic trends emerge from the arrangement of elements in the periodic table, and these trends are critical for understanding the chemical behavior of elements.
There are a few factors that commonly affect the atomic and physical properties of elements, such as atomic and ionic radius, electronegativity, and ionisation energy.
The net attractive force of the nucleus on the valence electrons, also termed as effective nuclear charge, Zeff, depends on both the nuclear charge and the screening effect.
Zeff = Z – screening effect
Let’s see how we can apply these factors to explain atomic and physical properties of elements across the periodic table.
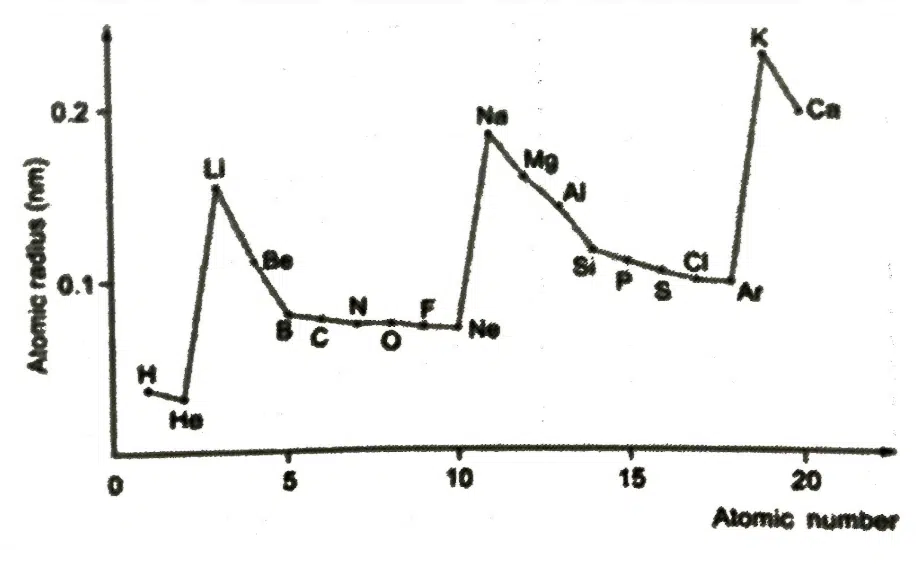
Down the group | Across the period |
↑ number of protons, ↑ nuclear charge (Z) | ↑ number of protons, ↑ Z |
↑ number of inner shell of electrons, ↑ shielding effect | Number of inner shell of electrons is constant, shielding effect relatively constant |
↑ screening effect outweighs ↑ Z as valence electrons are further away from the nucleus | ↑ effective Z |
↓ effective Z | |
↓ strength of electrostatic attraction between nucleus and valence electrons | ↑ strength of electrostatic attraction between nucleus and valence electrons |
↑ atomic radius | ↓ atomic radius |
Electronegativity is the relative tendency of an atom to attract electrons in a covalent bond.
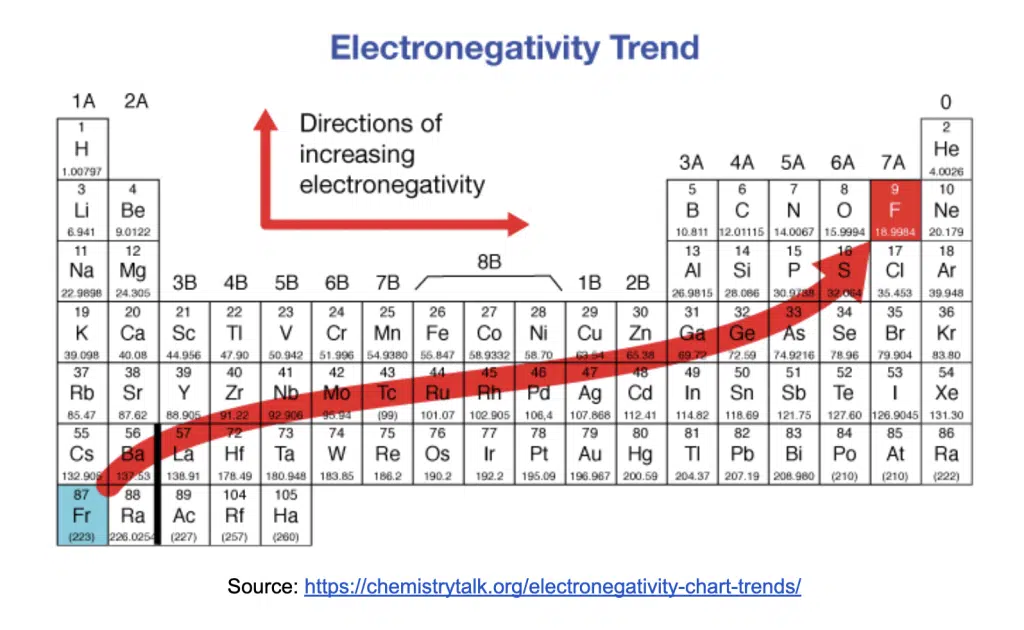
Down the group | Across the period |
↑ number of protons, ↑ Z | ↑ number of protons, ↑ Z |
↑ number of inner shell of electrons, ↑ shielding effect | Number of inner shell of electrons is constant, shielding effect relatively constant |
↑ screening effect outweighs ↑ Z as valence electrons are further away from the nucleus | ↑ effective Z |
↓ effective Z | |
↓ strength of electrostatic attraction between nucleus and valence electrons | ↑ strength of electrostatic attraction between nucleus and valence electrons |
↑ atomic radius | ↓ atomic radius |
↓ tendency to attract a bonding pair of electrons | ↑ tendency to attract a bonding pair of electrons |
↓ electronegativity | ↑ electronegativity |
Down the group | Across the period |
↑ number of protons, ↑ Z | ↑ number of protons, ↑ Z |
↑ number of inner shell of electrons, ↑ shielding effect | Number of inner shell of electrons is constant, shielding effect relatively constant |
↑ screening effect outweighs ↑ Z as valence electrons are further away from the nucleus | ↑ effective Z |
↓ effective Z | |
↑ atomic radius | ↓ atomic radius |
↓ strength of electrostatic attraction between nucleus and valence electrons | ↑ strength of electrostatic attraction between nucleus and valence electrons |
↓ energy required to remove valence electrons | ↑ energy required to remove valence electrons |
↓ 1st IE | ↑ 1st IE |
Take note of the following abnormalities: From Group 2 to 13 (e.g., Be to B – 1s22s2 to 1s22s22p1) Less energy required to remove a 2p electron than a 2s electron as the 2p electron is further from nucleus From Group 15 to 16 (e.g., N to O – 1s22s22p3 to 1s22s22p4) Less energy required to remove an electron from paired electron than unpaired electron due to electrostatic repulsion between paired electrons |
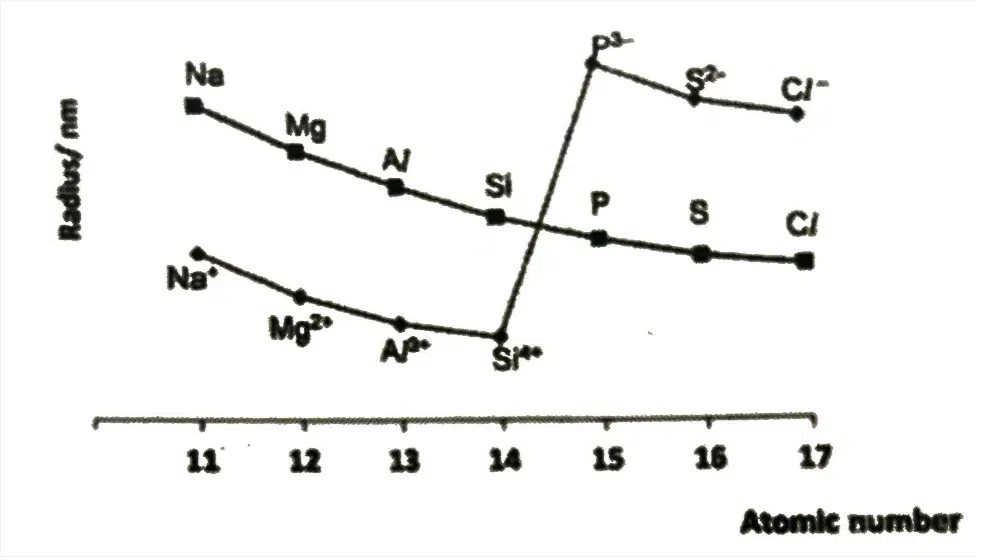
Cation | Anion |
Atoms lose some/all valence electrons to form cations | Electrons added to outermost quantum shell of atoms to form anions |
Cations have 1 less quantum shell of electrons than neutral atom | Increase electron-electon repulsion in outermost quantum shell of electrons |
Ionic radii of cations SMALLER than atomic radii of corresponding atoms | Ionic radii of anions LARGER than atomic radii of corresponding atoms |
Isoelectronic Series (e.g., Na+ to Si4+, P3- to Cl-)
↑ number of protons, ↑ Z
Number of inner shell of electrons is constant, shielding effect relatively constant
↑ effective Z
↑ strength of electrostatic attraction between nucleus and valence electrons
↓ ionic radius
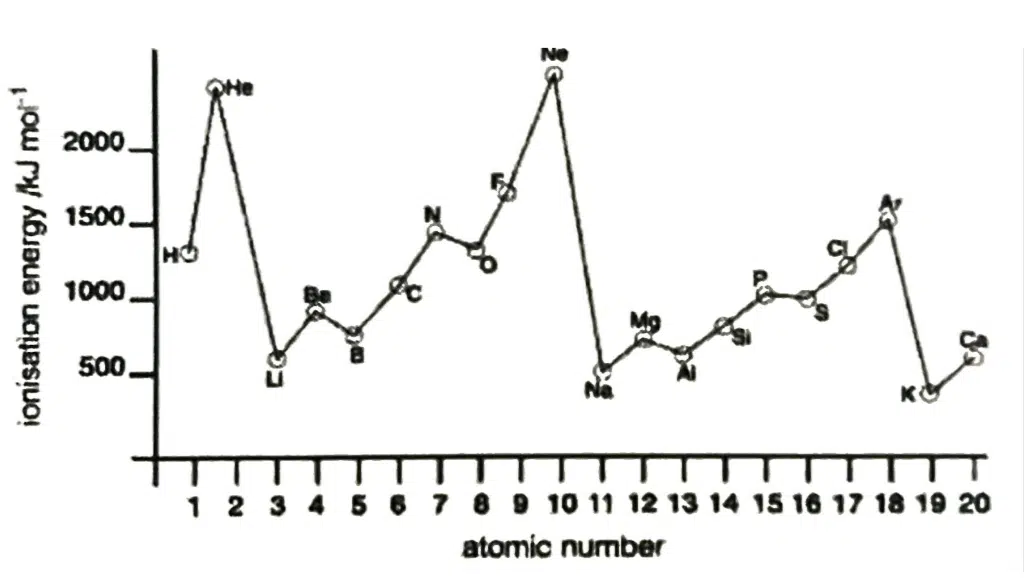
First Ionisation Energy, IE
First Ionization Energy is defined as the amount of energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous singly charged positive ions.
X(g) → X+(g) + e−
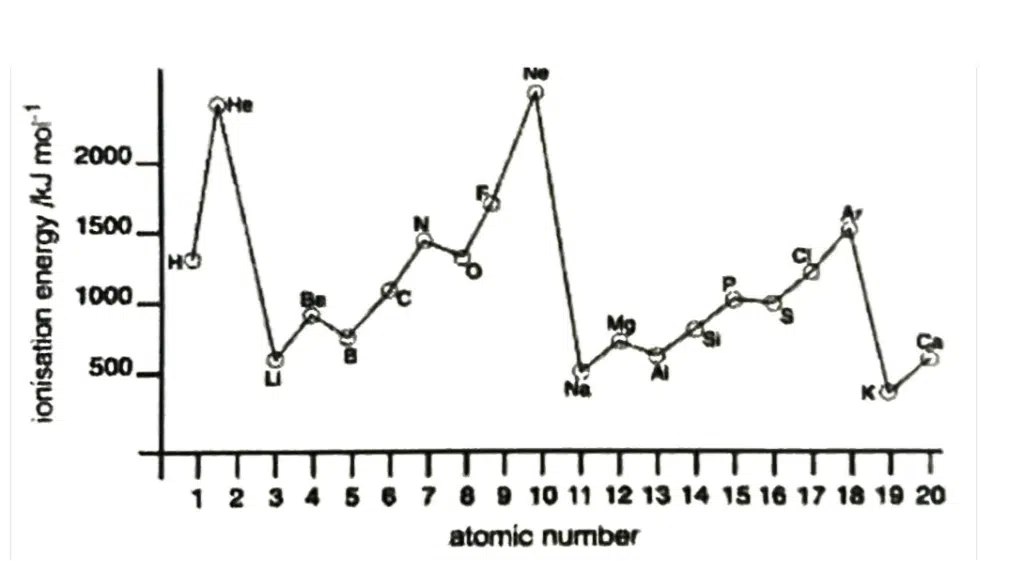
Down the group | Across the period |
↑ number of protons, ↑ Z | ↑ number of protons, ↑ Z |
↑ number of inner shell of electrons, ↑ shielding effect | Number of inner shell of electrons is constant, shielding effect relatively constant |
↑ screening effect outweighs ↑ Z as valence electrons are further away from the nucleus | ↑ effective Z |
↓ effective Z | |
↑ atomic radius | ↓ atomic radius |
↓ strength of electrostatic attraction between nucleus and valence electrons | ↑ strength of electrostatic attraction between nucleus and valence electrons |
↓ energy required to remove valence electrons | ↑ energy required to remove valence electrons |
↓ 1st IE | ↑ 1st IE |
Take note of the following abnormalities: From Group 2 to 13 (e.g., Be to B – 1s22s2 to 1s22s22p1) Less energy required to remove a 2p electron than a 2s electron as the 2p electron is further from nucleus From Group 15 to 16 (e.g., N to O – 1s22s22p3 to 1s22s22p4) Less energy required to remove an electron from paired electron than unpaired electron due to electrostatic repulsion between paired electrons |
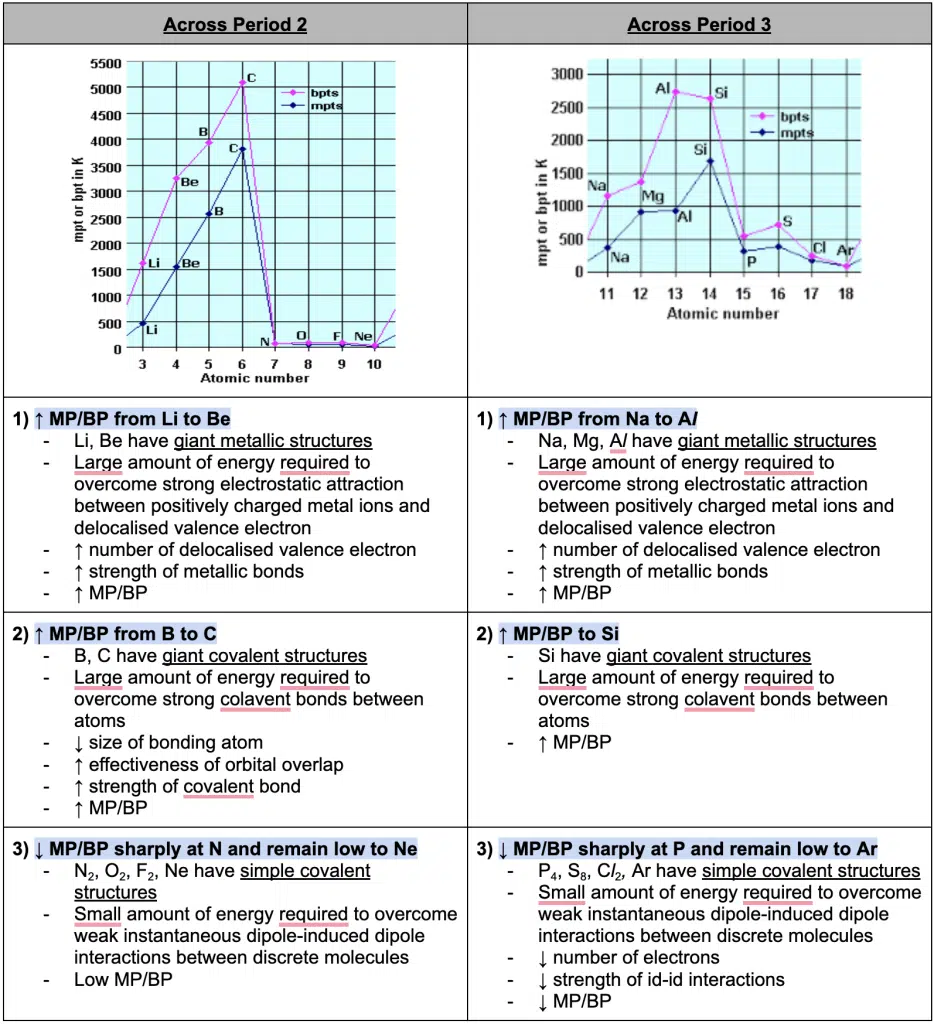
Melting and Boiling Point
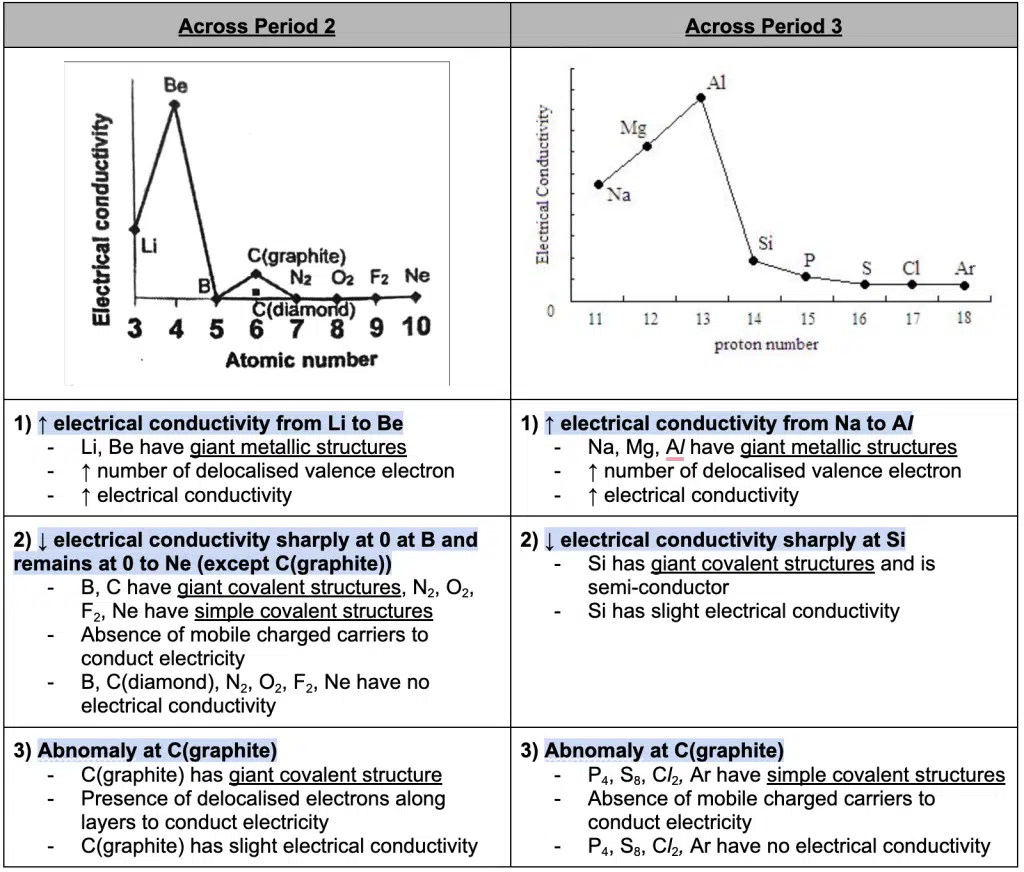
Electrical Conductivity
Volatility is the tendency to vapourise. It depends on the boiling point of substance.
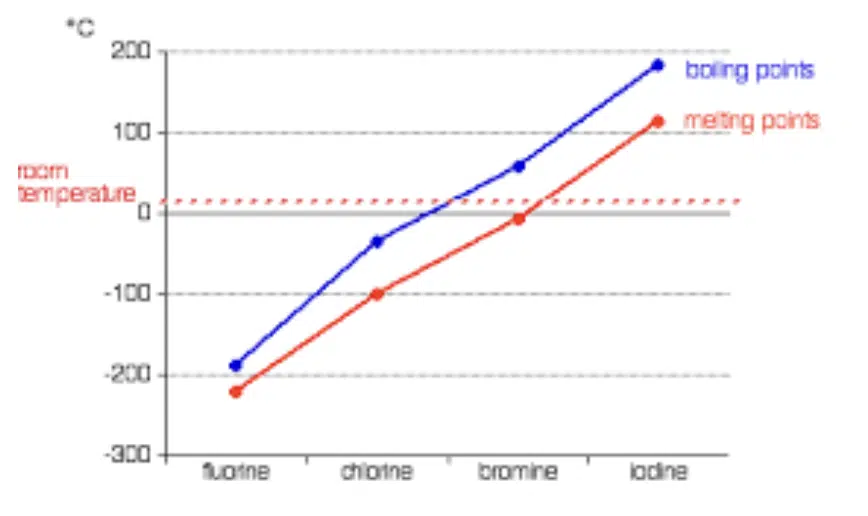
Down Group 17
Halogens have simple covalent structure
(exist as discrete molecules held together by weak instantaneous dipole-induced dipole interactions)
↑ number of electrons
↑ strength of instantaneous dipole-induced dipole interactions
↑ energy required to overcome interactions
↑ BP
↓ volatility
Apart from atomic and physical properties, there are also trends and variations in the chemical properties of the elements across the Periodic Table. Particularly, we are interested in the Period 3, Group 2, and Group 17 elements.
Chloride | NaCl | MgCl2 | AlCl3 | SiCl4 | PCl5 |
Chemical bonding | Ionic | Ionic (with covalent character) | Covalent (with ionic character) | Covalent | |
Structure | Giant ionic | Simple covalent | |||
MP/BP | High | Low | |||
Oxidation Number | +1 | +2 | +3 | +4 | +5 |
Chloride | Reaction with Water | pH | |
NaCl |
| Hydration: NaCl(s) → Na+(aq) + Cl–(aq) | 7 |
MgCl2 |
| Hydration: MgCl2(s) + 6H2O(i) → [Mg(H2O)6]2+(aq) + 2Cl–(aq) Hydrolysis: [Mg(H2O)6]2+(aq) + H2O(l) [Mg(H2O)5(OH)]+(aq) + H3O+(aq) | 6.5 |
AlCl3 |
| Hydration: AlCl3(s) + 6H2O(l) → [Al(H2O)6]3+(aq) + 3Cl–(aq) Hydrolysis: [Al(H2O)6]3+(aq) + H2O(l) [Al(H2O)5(OH)]2+(aq) + H3O+(aq) | 3 |
SiCl4 |
| Hydrolysis: SiCl4(l) + 2H2O(l) → SiO2(s) + 4HCl(aq) | 2 |
PCl5 |
| Hydrolysis: PCl5(l) + 4H2O(l) → H3PO4(s) + 5HCl(aq) | 2 |
Oxide | Na2O | MgO | Al2O3 | SiO2 | P4O10 | SO3 |
Chemical bonding | Ionic | Ionic (with covalent character) | Covalent | |||
Structure | Giant ionic | Giant covalent | Simple covalent | |||
MP/BP | High | Low | ||||
Type of oxides | Basic | Amphoteric | Acidic | |||
Oxide | Reaction with Water | pH | |
Na2O | Readily dissolves in water to give alkaline solution | Na2O(s) + H2O(l) → 2NaOH(aq) | 13 |
MgO | Dissolves slightly in water to give a weak alkaline solution | MgO(s) + H2O(l) Mg(OH)2(aq) | 9 |
Al2O3 | Insoluble in water due to high lattice energy | – | – |
SiO2 | Insoluble in water due to high lattice energy | – | – |
P4O10 | Readily dissolves in water to give an acidic solution | P4O10(s) + 6H2O(l) → 4H3PO4(aq) | 2 |
SO3 | Readily dissolves in water to give an acidic solution | SO3(g) + H2O(l) → H2SO4(aq) | 2 |
Oxide | Reaction with Acid/Base | |
Na2O |
| Na2O(s) + 2HCl(aq) → 2NaCl(aq) + H2O(l) |
MgO |
| MgO(s) + 2HCl(aq) → MgCl2(aq) + H2O(l) |
Al2O3 |
| Al2O3(s) + 6HCl(aq) → 2AlCl3(aq) + 3H2O(l) Al2O3(s) + 2NaOH(aq) + 3H2O(l) → 2NaAl(OH)4(aq) |
SiO2 |
| SiO2(s) + 2NaOH(aq) → Na2SiO3(aq) + H2O(l) |
P4O10 |
| P4O10(s) + 12NaOH(aq) → 4Na3PO4(aq) + 6H2O(l) |
SO3 |
| SO3(g) + 2NaOH(aq) → Na2SO4(aq) + H2O(l) |
Chemical Property | Trend | Explanation |
Reducing Power | Increases down the group |
|
Thermal Stability of Group 2 Compounds | Increases down the group |
|
Chemical Property | Trend | Explanation |
Oxidising Power | Decreases down the group |
|
Thermal Stability of Hydrogen halides | Decreases down the group |
|
‘Periodic Table’ is a heavy topic in ‘A’ Levels H2 Chemistry, incorporating content from across different topics. However, it is definitely not difficult once you are able to understand the main group and period patterns associated with the periodic table. Do remember to practice with examples and regularly revisit periodic trends to reinforce your understanding. Best of luck, and may your enthusiasm for chemistry continue to inspire you in all your future pursuits!
Find this helpful? Find out more by joining us at Science of Studying!
Prepared by: Tan Wee Leng
This article is written based on the 9729 Chemistry GCE Advanced Level H2 Syllabus. https://www.seab.gov.sg/docs/default-source/national-examinations/syllabus/alevel/2024syllabus/9729_y24_sy.pdf
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us