- 8808 2348
- blacklord1234@gmail.com
- Mon - Sun 9:00AM - 5:00PM

The periodic table is a cornerstone of chemistry, providing a comprehensive map of the elements and their properties. For O Level Pure Chemistry students, mastering the periodic table is essential. There are three subtopics that will be tested in exams – periodic trends, group properties, and transition elements.
Here’s a detailed guide to help you understand and effectively use the periodic table.
Click here for A Levels Period Table Guide.
But before you go on reading… You might want to download a pdf copy of this article as it is quite long!
Click the ‘Download’ button, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)

The Periodic Table is an arrangement of elements in the order of increasing proton (atomic) number.
A horizontal row is called a period.
A vertical column is called a group.
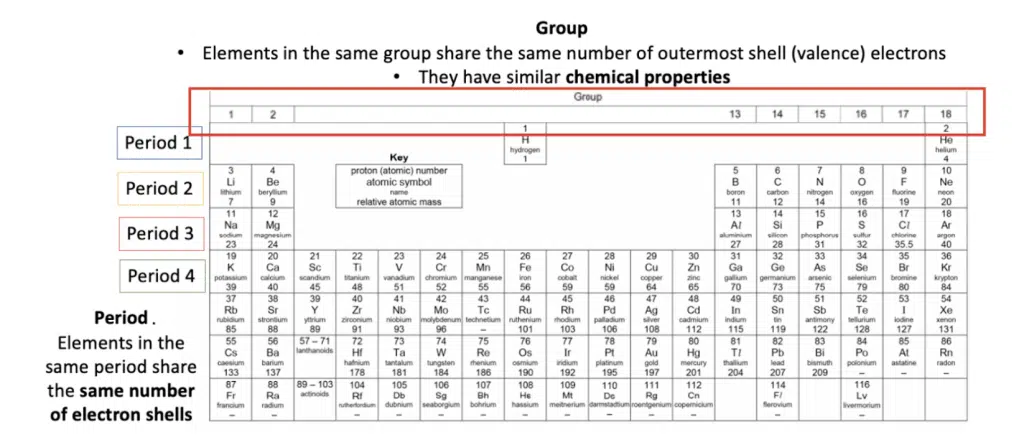
There are three main classes of elements:
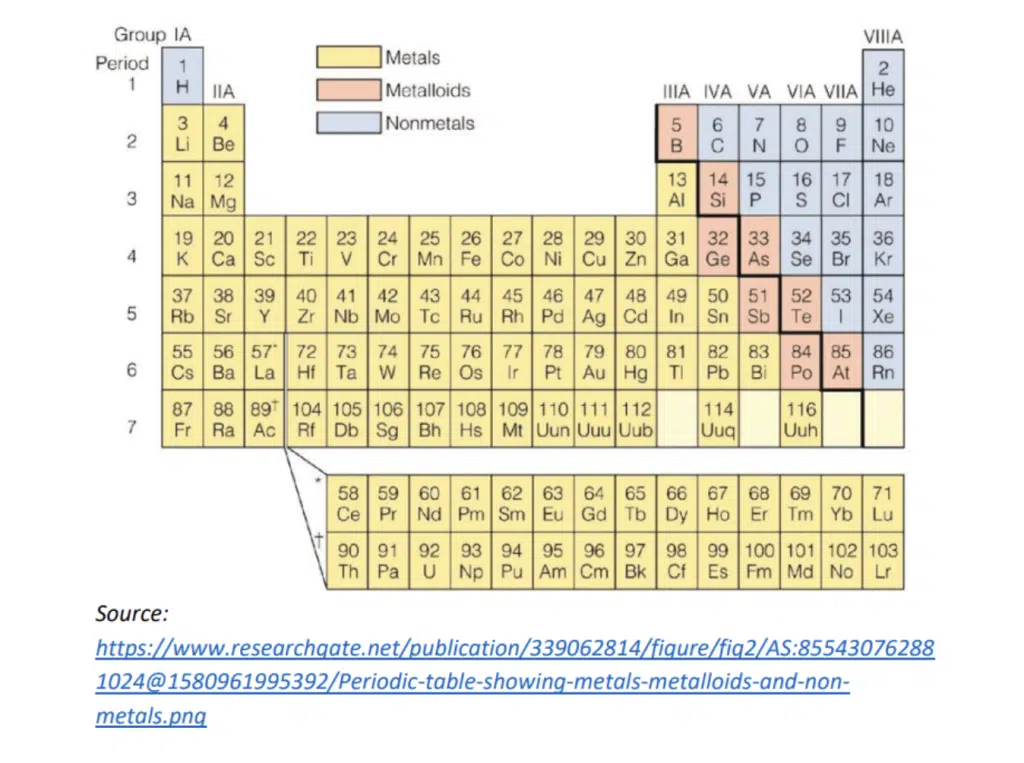
There are three groups of elements that we are particularly interested in.
Group 1 elements, also known as alkali metals, are characterised by having only 1 valence electron in the valence shell. This singular electron defines many of their physical and chemical properties.
Physical Properties of Alkali Metals
Chemical Properties of Alkali Metals
Types of reactions:
Reaction | Products |
React with non-metals | Form ionic compounds |
React with water | Form metal hydroxide and hydrogen gas |
React with acid | Form salt and hydrogen gas |
React with chlorine or air (O2) | Form metal chloride or metal oxide |
Trends of Alkali Metals
Trend | Explanations | |
Melting / Boiling Point | Decreases | ↑ number of electron shell, ↑ atomic radius, ↓ attractive forces between delocalised electrons and nucleus, ↓ energy needed to overcome metallic bond |
Density | Increases | |
Reactivity | Increases | ↑ number of electron shell, ↑ atomic radius, ↓ attractive forces between valence electron and nucleus, easier to lose electron to form cation |
Halogens, found in Group 17 of the periodic table, are a fascinating group of non-metals known for their high reactivity and distinctive properties. With 7 valence electrons in the valence shell, halogens are just one electron short of achieving a stable noble gas configuration. As such, they exists as diatomic molecule (e.g., F2) to achieve electronic stability.
Physical Properties of Halogens
Chemical Properties of Halogens
Types of reactions:
Reaction | Products |
React with metals | Form ionic compounds (metal halide) |
React with another non-metals | Form covalent compounds |
Trends of Halogens
Trend | Explanations | |
Melting / Boiling Point | Increases | ↑ number of electron shell, ↑ number of electrons, ↑ strength of intermolecular forces of attraction between molecules, ↑ energy needed to overcome attractive force |
Density | Increases | |
Reactivity | Decreases | ↑ number of electron shell, ↑ atomic radius, ↓ attractive forces between valence electron and nucleus, harder to gain electron to form anion |
Halogen Displacement
Halogen displacement reactions are a key concept in understanding the reactivity trends within Group 17 elements. These reaction occur when a more reactive halogen can displace the less reactive halide ion from its compound or solution.
Reactivity Order:
The reactivity of halogens decreases down Group 17, with fluorine being the most reactive, followed by chlorine, bromine, and iodine (F > Cl > Br > I). For example, when chlorine gas (Cl2) is added to a solution containing bromide ions (Br–), the chlorine will displace the bromine, forming chloride ions (Cl–) and free bromine (Br2):
Cl2 + 2Br− → 2Cl− + Br2
Observation of Displacement:
The identity of halogens during halogen displacement can be easily inferred from its colour and physical state as shown below.
F2 | Cl2 | Br2 | I2 | |
Colour | Pale yellow | Pale-greenish yellow | Reddish-brown | Black |
Physical state at r.t.p | Gas | Gas | Liquid | Solid |
Noble gases, found in Group 18 of a periodic table, are unique due to their fully filled valence electron shells. This configuration allows them to exist as monatomic atoms (e.g., Ar, Ne) and makes them extremely stable and largely unreactive. Here’s a closer look at their properties and uses:
Physical Properties of Noble Gases
Chemistry Properties of Noble Gases
Uses of Noble Gases
Noble gases are widely used due to their lack of reactivity. For example,
Transition elements, located in the central block of the Periodic Table (between Group 2 and 13), are metals that exhibit unique properties compared to the main group elements. These elements include iron, copper, nickel, and many others.
Understanding the periodic table is essential for mastering O-Level Chemistry. These concepts not only provide the foundation for other chemistry topics, but also help in making sense of the chemical reactions and properties observed in everyday life. Keep exploring, questioning, and connecting the dots—this is the essence of learning chemistry.
Find this helpful? Find out more by joining us at Science of Studying!
Prepared by: Tan Wee Leng
This article is written based on the 6092 Chemistry GCE Ordinary Level Syllabus. https://www.seab.gov.sg/docs/default-source/national-examinations/syllabus/olevel/2024syllabus/6092_y24_sy.pdf
You might want to download a pdf copy of this article for future reference!
Click the white download button below, enter your email, and the pdf file will be delivered to your inbox! (Remember to check spam!)
The Science of Studying provides live online tuition via Zoom classes for Combined/Pure Chemistry, Biology, and Physics. To date, we have taught 800+ students over 12 years.
In case you are wondering, yes – there is a science behind studying!
At Science of Studying, we use our SOS system™ to teach our classes so that even last-minute students can see remarkable improvements in their grades – without mind-numbing memorisation of textbooks and without the drudgery of doing numerous assessment books.
All these conducted in a fun, interactive, stress-free online environment.
If you need help with your Chemistry, Biology, and Physics subjects, do reach out to us and we will see what we can do to help.
Contact Us: Click Here
Admin number: +65 88082348

The SOS system™️ guides students through an effective process of:
Join our proven online tuition programs and see real improvements in understanding, confidence, and school results.
Book a free trial lesson and start the journey today or discover more below:
WhatsApp us